To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Battery (electricity)
A battery is a device made of one or more electrochemical cells, which store chemical energy and make it available in an electrical form. There are many types of electrochemical cells, including galvanic cells, electrolytic cells, fuel cells, flow cells, and voltaic cells.[1] Formally, an electrical "battery" is an array of similar voltaic cells ("cells") connected in series. However, in many contexts it is common to call a single cell a battery.[2] A battery's characteristics may vary due to many factors including internal chemistry, current drain, and temperature. There are two types of batteries, primary (disposable) and secondary (rechargeable), both of which convert chemical energy to electrical energy. Primary batteries can only be used once because they use up their chemicals in an irreversible reaction. Secondary batteries can be recharged because the chemical reactions they use are reversible; they are recharged by running a charging current through the battery, but in the opposite direction of the discharge current.[3] Rechargeable batteries can be charged hundreds of times before wearing out; and even after wearing out some can be recycled. Although an early form of battery may have been used in antiquity, the modern development of batteries started with the Voltaic pile, invented by the Italian physicist Alessandro Volta in 1800. Since then, batteries have gained popularity as they became portable and useful for many purposes.[4] The widespread use of batteries has created many environmental concerns, such as toxic metal pollution.[5] Many reclamation companies recycle batteries to reduce the number of batteries going into landfills.[6] Generally, battery life can be prolonged by storing the battery in a cool place and using it at an appropriate current.[7] Additional recommended knowledge
HistoryA controversial claim is that the first known artifacts that may have served as batteries are the Baghdad Batteries, which existed some time between 250 BC and 640 AD. The modern story of the battery begins in the 1780s with the discovery of "animal electricity" by Luigi Galvani, which he published in 1791.[8] He created an electric circuit consisting of two different metals, with one touching a frog's leg and the other touching both the leg and the first metal, thus closing the circuit. In modern terms, the frog's leg served as both electrolyte and detector, and the metals served as electrodes. He noticed that even though the frog was dead, its legs would twitch when he touched them with the metals.[9] By 1791, Alessandro Volta realized that the frog's moist tissues could be replaced by cardboard soaked in salt water, and the frog's muscular response could be replaced by another form of electrical detection. He already had studied the electrostatic phenomenon of capacitance, which required measurements of electric charge and of electrical potential. Building on this experience Volta was able to detect electric current flow through his system, now called a voltaic cell, or cell for short. The terminal voltage of a cell that is not discharging is called its electromotive force (emf), and has the same unit as electrical potential, named (voltage) and measured in volts, in honor of Volta. In 1799, Volta invented the battery by placing many voltaic cells in series, literally piling them one above the other. This Voltaic Pile gave a greatly enhanced net emf for the combination,[10] with a voltage of about 50 volts for a 32-cell pile.[11] In many parts of Europe batteries continue to be called piles. Unfortunately, Volta did not appreciate that the voltage was due to chemical reactions. He thought that his cells were an inexhaustible source of energy, and that the associated chemical effects (e.g., corrosion) were a mere nuisance, rather than, as Michael Faraday showed around 1830, an unavoidable consequence of their operation. Banks of cells came to be called 'batteries', presumably after the earlier use by Benjamin Franklin to describe Leyden jars (capacitors) in series and in parallel, which vaguely resembled artillery batteries.[12] Although early batteries were of great value for experimental purposes, their limitations made them impractical for large current drain. Later, starting with the Daniell cell in 1836, batteries provided more reliable currents and were adopted by industry for use in stationary devices, particularly in telegraph networks where they were the only practical source of electricity, since electrical distribution networks did not exist then.[13] These wet cells used liquid electrolytes, which were prone to leaks and spillage if not handled correctly. Many used glass jars to hold their components, which made them fragile. These characteristics made wet cells unsuitable for portable appliances. Near the end of the 19th century, the invention of dry cell batteries, which replaced liquid electrolyte with a paste made portable electrical devices practical. The battery has since become a common power source for many household and industrial applications. According to a 2005 estimate, the worldwide battery industry generates US$48 billion in sales annually.[14] How batteries workA battery is a device that converts chemical energy directly to electrical energy.[15] It consists of one or more voltaic cells. Each voltaic cell consists of two half cells connected in series by a conductive electrolyte. One half-cell is the positive electrode (cathode), and the other is the negative electrode (anode). The electrodes do not touch each other but are electrically connected by the electrolyte, which can be either solid or liquid.[16] In many cells the materials are enclosed in a container, and a separator, which is porous to the electrolyte, prevents the electrodes from coming into contact. Each half cell has an electromotive force (or emf), with the net emf of the battery being the difference between the emfs of the half-cells, a fact first recognized by Volta. Thus, if the electrodes have emfs The electrical potential difference, or The voltage developed across a cell's terminals depends on the chemicals used in it and their concentrations. For example, alkaline and carbon-zinc cells both measure about 1.5 volts, due to the energy release of the associated chemical reactions. Because of the high electrochemical potential changes in the reactions of lithium compounds, lithium cells can provide as much as 3 volts or more. Classification of batteriesDisposable and rechargeable
Batteries are usually divided into two broad classes:
Historically, some types of primary batteries used, for example, for telegraph circuits, were restored to operation by replacing the components of the battery consumed by the chemical reaction. Secondary batteries are not indefinitely rechargeable due to dissipation of the active materials, loss of electrolyte, and internal corrosion. From a user's viewpoint, at least, batteries can be generally divided into two main types: non-rechargeable (disposable) and rechargeable. Each is in wide usage. Disposable batteries are also called primary cells, are intended to be used once and discarded. These are most commonly used in portable devices with either low current drain, only used intermittently, or used well away from an alternative power source. Primary cells were also commonly used for alarm and communication circuits where other electric power was only intermittently available. Primary cells cannot be reliably recharged, since the chemical reactions are not easily reversible and active materials may not return to their original forms. Battery manufacturers recommend against attempting to recharge primary cells, although some electronics enthusiasts claim it is possible to do so using a special type of charger.[21] By contrast, rechargeable batteries or secondary cells can be re-charged by applying electrical current, which reverses the chemical reactions that occur in use. Devices to supply the appropriate current are called chargers or rechargers. The oldest form of rechargeable battery still in modern usage is the "wet cell" lead-acid battery.[22] This battery is notable in that it contains a liquid in an unsealed container, requiring that the battery be kept upright and the area be well ventilated to ensure safe dispersal of the hydrogen gas produced by these batteries during overcharging. The lead-acid battery is also very heavy for the amount of electrical energy it can supply. Despite this, its low manufacturing cost and its high surge current levels make its use common where a large capacity (over approximately 10Ah) is required or where the weight and ease of handling are not concerns. A common form of lead-acid battery is the modern wet-cell car battery. This can deliver about 10,000 watts of power for a short period, and has a peak current output that varies from 450 to 1100 amperes. An improved type of liquid electrolyte battery is the sealed valve regulated lead acid (VRLA) battery, popular in automotive industry as a replacement for the lead-acid wet cell, as well as in many lower capacity roles including smaller vehicles and stationary applications such as emergency lighting and alarm systems. The one-way pressure activated valve eliminates electrolyte evaporation while allowing out-gassing to prevent rupture. This greatly improves resistance to damage from vibration and heat. VRLA batteries have the electrolyte immobilized, usually by one of two means:
Other portable rechargeable batteries include several "dry cell" types, which are sealed units and are therefore useful in appliances like mobile phones and laptops. Cells of this type (in order of increasing power density and cost) include nickel-cadmium (NiCd), nickel metal hydride (NiMH), and lithium-ion (Li-Ion) cells. Recent developments include AA Batteries with embedded functionality such as USBCELL[23] with built-in charger and USB connector within the AA format, enabling the battery to be charged by plugging into a USB port without a charger, and LSD - Low self discharge NiMh mix chemistries such as Hybrio, Recyko, Eneloop where cells are precharged prior to shipping (within UN Proposition 304 shipping legislation that limits charge density on shipments) DisposableNot designed to be rechargeable - sometimes called "primary cells". "Disposable" may also imply that special disposal procedures must take place for proper disposal according to regulation, depending on battery type.
Rechargeable
Also known as secondary batteries or accumulators. The National Electrical Manufacturers Association has estimated that U.S. demand for rechargeables is growing twice as fast as demand for non-rechargeables. [26] There are a few main types:
Flow batteriesFlow batteries are a special class of rechargeable battery where additional quantities of electrolyte are stored outside the main power cell of the battery, and circulated through it by pumps or by movement.[29] Flow batteries can have extremely large capacities and are used in marine applications and are gaining popularity in grid energy storage applications. Zinc-bromine[29] and vanadium redox batteries are typical examples of commercially available flow batteries. Homemade cellsAlmost any liquid or moist object that has enough ions to be electrically conductive can serve as the electrolyte for a cell. As a novelty or science demonstration, it is possible to insert two electrodes made of different metals into a lemon,[30] potato,[31] et cetera and generate small amounts of electricity. "Two-potato clocks" are also widely available in hobby and toy stores; they consist of a pair of cells, each consisting of a potato (lemon, et cetera) with two electrodes inserted into it, wired in series to form a battery with enough voltage to power a digital clock.[32] Homemade cells of this kind are of no real practical use, because they produce far less current—and cost far more per unit of energy generated—than commercial cells, due to the need for frequent replacement of the fruit or vegetable. In addition, one can make a voltaic pile from two coins (such as a nickel and a penny) and a piece of paper towel dipped in salt water. Such a pile would make very little voltage itself, but when many of them are stacked together in series, they can replace normal batteries for a short amount of time.[33] Sony has developed a biologically friendly battery that generates electricity from sugar in a way that's similar to what's found in living organisms. The battery generates electricity through the use of enzymes that break down carbohydrates, which are essentially sugar.[34] Lead acid cells can easily be manufactured at home, but a tedious charge/discharge cycle is needed to 'form' the plates. This is a process whereby lead sulfate forms on the plates, and during charge is converted to lead dioxide (positive plate) and pure lead (negative plate). Repeating this process results in a microscopically rough surface, with far greater surface area being exposed. This increases the current the cell can deliver. For an example, see [2]. Daniel cells are also easy to make at home. Aluminum-air batteries can also be produced. By size and use
Battery packsThe cells in a battery can be connected in parallel, series, or in both. A parallel combination of cells has the same voltage as a single cell, but can supply a higher current (the sum of the currents from all the cells). A series combination has the same current rating as a single cell but its voltage is the sum of the voltages of all the cells. Most practical electrochemical batteries, such as 9-volt flashlight (torch) batteries and 12-volt automobile (car) batteries, have several cells connected in series inside the casing.[35] Parallel arrangements suffer from the problem that, if one cell discharges faster than its neighbour, current will flow from the full cell to the empty cell, wasting power and possibly causing overheating. Even worse, if one cell becomes short-circuited due to an internal fault, its neighbour will be forced to discharge its maximum current into the faulty cell, leading to overheating and possibly explosion.[36] Cells in parallel are therefore usually fitted with an electronic circuit to protect them against these problems. In both series and parallel types, the energy stored in the battery is equal to the sum of the energies stored in all the cells. Traction batteriesTraction batteries are high-power batteries designed to provide propulsion to move a vehicle, such as an electric car or tow motor. A major design consideration is power to weight ratio since the vehicle must carry the battery.[37] While conventional lead acid batteries[38] with liquid electrolyte have been used, gelled electrolyte[39] and AGM-type[40] can also be used, especially in smaller sizes. The largest installations of batteries for propulsion of vehicles are found in submarines, although the toxic gas produced by seawater contact with acid electrolyte is a considerable hazard. Battery types commercially used in electric vehicles include
See also: battery electric vehicles and hydrogen vehicle. Battery capacity and dischargingThe more electrolyte and electrode material there is in the cell, the greater the capacity of the cell. Thus a small cell has less capacity than a larger cell, given the same chemistry (e.g. alkaline cells), though they develop the same open-circuit voltage.[41] Because of the chemical reactions within the cells, the capacity of a battery depends on the discharge conditions such as the magnitude of the current, the duration of the current, the allowable terminal voltage of the battery, temperature, and other factors.[41] The available capacity of a battery depends upon the rate at which it is discharged.[42] If a battery is discharged at a relatively high rate, the available capacity will be lower than expected. The battery capacity (C) that battery manufacturers print on a battery is the product of 20 hours multiplied by the maximum constant current that a new battery can supply for 20 hours at 68 F (20 C)[3], down to a predetermined terminal voltage per cell. A battery rated at 100 A·h will deliver 5 A over a 20 hour period at room temperature. However, if it is instead discharged at 50 A, it will run out of charge before the theoretically expected 2 hours. For this reason, a battery capacity rating is always related to an expected discharge duration -- the standard duration is 20 hours. where
Ampere-hours are commonly called amp-hours. In general, the higher the ampere-hour rating, the longer the battery will last for a certain load. Installing batteries with different A·h ratings will not affect the operation of a device rated for a specific voltage unless the load limits of the battery are exceeded. For example a 1.5V application that has a load impedance of 1.2 ohms is drawing 1.25A, 1.8W (ohms law) continuously can not be powered by a AAA alkaline type battery for any amount of time. Theoretically with a capacity of 1250mAh the AAA could drive 1.25A for 1 hour. Realistically the AAA is not capable of supplying that much continuous current to this load at 1.5V. When the battery limits are exceeded the voltage across the battery is pulled down. When the 1.5V AAA alkaline is placed in in parallel with the 1.2 ohm Load we measure only 890mA at 1V 0.9W, thus the demand of the circuit is not met by this battery.
Conversion to energyThe ampere-hour (A·h) rating of a battery is related to the amount of energy it stores when fully charged.[44] If two batteries have the same nominal voltage, then the one with the higher A·h rating stores more energy. It also typically takes longer to recharge. Accurate calculation of the energy stored in the battery is complex due to the varying terminal voltage during discharge (and so the need to specify an end terminal voltage for discharge), and due to the dependency of total capacity on the discharge rate and discharge duty. Secondary batteries always yield less energy than was used to charge them, since (among other reasons) the terminal voltage during charging is higher than during discharging. The nickel cadmium and nickel metal-hydride designs have efficiencies of around 66%.[45] However, modern lithium designs have almost negated this wastage as they can have efficiencies of around 99%.[46] Effect of a battery's internal resistanceA battery can be simply modeled as a perfect voltage source (i.e. one with zero internal resistance) in series with a resistor. The voltage source depends mainly on the chemistry of the battery, not on whether it is empty or full. When a battery runs down, its internal resistance increases. When the battery is connected to a load (e.g. a light bulb), which has its own resistance, the resulting voltage across the load depends on the ratio of the battery's internal resistance to the resistance of the load. When the battery is fresh, its internal resistance is low, so the voltage across the load is almost equal to that of the battery's internal voltage source. As the battery runs down and its internal resistance increases, the voltage drop across its internal resistance increases, so the voltage at its terminals decreases, and the battery's ability to deliver power to the load decreases. The formula for calculating the voltage Vt at the terminals of a battery is where
This can be rearranged to calculate the internal resistance given the other quantities: Battery lifetimeLife of primary batteriesEven if never taken out of the original package, disposable (or "primary") batteries can lose 8 to 20 percent of their original charge every year at a temperature of about 20°–30°C.[48] This is known as the "self discharge" rate and is due to non-current-producing "side" chemical reactions, which occur within the cell even if no load is applied to it. The rate of the side reactions is reduced if the batteries are stored at low temperature, although some batteries can be damaged by freezing. High or low temperatures may reduce battery performance. This will affect the initial voltage of the battery. For an AA alkaline battery this initial voltage is approximately normally distributed around 1.6 volts. Life of rechargeable batteriesRechargeable batteries traditionally self-discharge more rapidly than disposable alkaline batteries; up to three percent a day (depending on temperature). However, modern Lithium designs have reduced the self-discharge rate to a relatively low level (but still poorer than for primary batteries). Due to their poor shelf life, rechargeable batteries should not be stored and then relied upon to power flashlights or radios in an emergency. For this reason, it is a good idea to keep alkaline batteries on hand. NiCd Batteries are almost always "dead" when purchased, and must be charged before first use. Although rechargeable batteries may be refreshed by charging, they still suffer degradation through usage. Low-capacity Nickel Metal Hydride (NiMH) batteries (1700-2000 mAh) can be charged for about 1000 cycles, whereas high capacity NiMH batteries (above 2500 mAh) can be charged for about 500 cycles.[49] Nickel Cadmium (NiCd) batteries tend to be rated for 1,000 cycles before their internal resistance increases beyond usable values. Normally a fast charge, rather than a slow overnight charge, will result in a shorter battery lifespan.[49] However, if the overnight charger is not "smart" (i.e. it cannot detect when the battery is fully charged), then overcharging is likely, which will damage the battery.[50] Degradation usually occurs because electrolyte migrates away from the electrodes or because active material falls off the electrodes. NiCd batteries suffer the drawback that they should be fully discharged before recharge. Without full discharge, crystals may build up on the electrodes, thus decreasing the active surface area and increasing internal resistance. This decreases battery capacity and causes the dreaded "memory effect". These electrode crystals can also penetrate the electrolyte separator, thereby causing shorts. NiMH, although similar in chemistry, does not suffer from ‘memory effect’ to quite this extent.[51] Automotive lead-acid rechargeable batteries have a much harder life. Because of vibration, shock, heat, cold, and sulfation of their lead plates, few automotive batteries last beyond six years of regular use. Automotive starting batteries have many thin plates to provide as much current as possible in a reasonably small package. Typically they are only drained a small amount before recharge. Care should be taken to avoid deep discharging a starting battery, since each charge and discharge cycle causes active material to be shed from the plates. Hole formation in the plates leads to less surface area for the current-producing chemical reactions, resulting in less available current when under load. Leaving a lead-acid battery in a deeply discharged state for any significant length of time allows the lead sulfate to crystallize, making it difficult or impossible to remove during the charging process. This can result in a permanent reduction in the available plate surface, and therefore reduced current output and energy capacity. "Deep-Cycle" lead-acid batteries such as those used in electric golf carts have much thicker plates to aid their longevity. The main benefit of the lead-acid battery is its low cost; the main drawbacks are its large size and weight for a given capacity and voltage. Lead-acid batteries should never be discharged to below 20% of their full capacity, because internal resistance will cause heat and damage when they are recharged. Deep-cycle lead-acid systems often use a low-charge warning light or a low-charge power cut-off switch to prevent the type of damage that will shorten the battery's life. Special "reserve" batteries intended for long storage in emergency equipment or munitions keep the electrolyte of the battery separate from the plates until the battery is activated, allowing the cells to be filled with the electrolyte. Shelf times for such batteries can be years or decades. However, their construction is more expensive than more common forms. Extending battery lifeBattery life can be extended by storing the batteries at a low temperature, as in a refrigerator or freezer, because the chemical reactions in the batteries are slower. Such storage can extend the life of alkaline batteries by ~5%; while the charge of rechargeable batteries can be extended from a few days up to several months.[52] In order to reach their maximum voltage, batteries must be returned to room temperature; therefore, alkaline battery manufacturers like Duracell do not recommend refrigerating or freezing batteries.[53] Battery heat, explosion and other hazardsA battery explosion is caused by the misuse or malfunction of a battery, such as attempting to recharge a primary (non-rechargeable) battery,[54] or short circuiting a battery.[55] With car batteries, explosions are most likely to occur when a short circuit generates very large currents. In addition, car batteries liberate hydrogen when they are overcharged (because of electrolysis of the water in the electrolyte). Normally the amount of overcharging is very small, as is the amount of explosive gas developed, and the gas dissipates quickly. However, when "jumping" a car battery, the high current can cause the rapid release of large volumes of hydrogen, which can be ignited by a nearby spark (for example, when removing the jumper cables). When a battery is recharged at an excessive rate, an explosive gas mixture of hydrogen and oxygen may be produced faster than it can escape from within the walls of the battery, leading to pressure build-up and the possibility of the battery case bursting. In extreme cases, the battery acid may spray violently from the casing of the battery and cause injury. Battery explosions can also occur in maintenance free lead-acid batteries if the valves fail by being blocked, for example. The pressure rises within the cells until a short-circuit ignites the hydrogen-oxygen mixture. Such explosions can cause severe personal injury. The problem can be detected in Car batteries if the sides appear at all swollen. Additionally, disposing of a battery in fire may cause an explosion as steam builds up within the sealed case of the battery.[55] Overcharging — that is, attempting to charge a battery beyond its electrical capacity — can also lead to a battery explosion, leakage, or irreversible damage to the battery. It may also cause damage to the charger or device in which the overcharged battery is later used. Short circuitA short circuit can lead to a battery fire or explosion. It often occurs when a battery is connected to itself, creating two points on a circuit with different potentials connected with zero or near-zero resistance.[56] The current of the circuit would be where Since 9-volt batteries have a male positive terminal and a female negative terminal, two such batteries could be "snapped" onto each other, creating 18 V with little resistance. Also, car lead-acid batteries can provide a lot of current for 12 V. Both types of batteries are particularly dangerous when shorted.[citation needed] Environmental considerationsSince their development over 250 years ago, batteries have remained among the most expensive energy sources, and their manufacturing consumes many valuable resources and often involves hazardous chemicals. Used batteries also contribute to electronic waste. For these reasons, many areas now have battery recycling services available to recover some of the more toxic (and sometimes valuable) materials from used batteries.[57] Batteries may be harmful or fatal if swallowed.[58] It is also important to prevent dangerous elements, such as lead, mercury, and cadmium, that are found in some types of batteries from entering the environment. GlossarySome common battery-related terms:
See also
References
Further reading
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Battery_(electricity)". A list of authors is available in Wikipedia. |





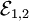 , then the net emf is
, then the net emf is 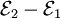 . (Hence, two identical electrodes and a common electrolyte give zero net emf.) Each half cell emf is due to a charge-transferring (or faradaic) chemical reaction at the electrode-electrolyte interface, which transfers charge across the interface. The reaction stops when the charge transfer is enough to cancel out the tendency of the reaction to occur. Non-charge-transferring, or nonfaradaic, reactions can also occur at the interface. These are undesirable, using up the chemicals without producing current (which is the rate of charge transfer). Additional, but relatively ineffective, faradaic reactions (also called parasitic or "side-reactions") can also occur.
. (Hence, two identical electrodes and a common electrolyte give zero net emf.) Each half cell emf is due to a charge-transferring (or faradaic) chemical reaction at the electrode-electrolyte interface, which transfers charge across the interface. The reaction stops when the charge transfer is enough to cancel out the tendency of the reaction to occur. Non-charge-transferring, or nonfaradaic, reactions can also occur at the interface. These are undesirable, using up the chemicals without producing current (which is the rate of charge transfer). Additional, but relatively ineffective, faradaic reactions (also called parasitic or "side-reactions") can also occur.
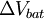 across the terminals of a battery is known as its terminal voltage, measured in
across the terminals of a battery is known as its terminal voltage, measured in 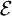 , meaning that to produce a potential difference of 1.5 V, chemical reactions inside would do 1.5
, meaning that to produce a potential difference of 1.5 V, chemical reactions inside would do 1.5 ![h_{[hours]} = \frac {C_{Batt [mAh]}} {I_{[mA]}}](images/math/a/b/6/ab607e0e7193063336ebd7959b4cd8b7.png)
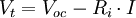
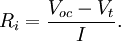
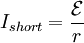
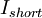 is the current,
is the current, 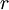 is the resistance. If an ideal battery (normal batteries have internal resistance) were shorted using an ideal wire, making the resistance 0, then
is the resistance. If an ideal battery (normal batteries have internal resistance) were shorted using an ideal wire, making the resistance 0, then 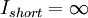 ,
,

