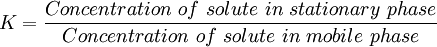To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Chromatography
Chromatography (from Greek χρώμα:chroma, colour and γραφειν:"grafein" to write) is the collective term for a family of laboratory techniques for the separation of mixtures. It involves passing a mixture dissolved in a "mobile phase" through a stationary phase, which separates the analyte to be measured from other molecules in the mixture and allows it to be isolated. Chromatography may be preparative or analytical. Preparative chromatography seeks to separate the components of a mixture for further use (and is thus a form of purification). Analytical chromatography normally operates with smaller amounts of material and seeks to measure the relative proportions of analytes in a mixture. The two are not mutually exclusive. Additional recommended knowledge
ExplanationAn analogy which is sometimes useful is to suppose a mixture of bees and wasps passing over a flower bed. The bees would be more attracted to the flowers than the wasps, and would become separated from them. If one were to observe at a point past the flower bed, the wasps would pass first, followed by the bees. In this analogy, the bees and wasps represent the analytes to be separated, the flowers represent the stationary phase, and the mobile phase could be thought of as the air. The key to the separation is the differing affinities among analyte, stationary phase, and mobile phase. The observer could represent the detector used in some forms of analytical chromatography. A key point is that the detector need not be capable of discriminating between the analytes, since they have become separated before passing the detector. HistoryIt was the Russian botanist Mikhail Semyonovich Tsvet who invented the first chromatography technique in 1900 during his research on chlorophyll. He used a liquid-adsorption column containing calcium carbonate to separate plant pigments. The method was described on December 30 1901 at the 11th Congress of Naturalists and Doctors (XI съезд естествоиспытателей и врачей) in St. Petersburg. The first printed description was in 1903, in the Proceedings of the Warsaw Society of Naturalists, section of biology. He first used the term chromatography in print in 1906 in his two papers about chlorophyll in the German botanical journal, Berichte der Deutschen Botanischen Gesellschaft. In 1907 he demonstrated his chromatograph for the German Botanical Society. Interestingly, Mikhail's surname "Цвет" means "color" in Russian, so there is the possibility that his naming the procedure chromatography (literally "color writing") was a way that he could make sure that he, a commoner in Tsarist Russia, could be immortalized. In 1952 Archer John Porter Martin and Richard Laurence Millington Synge were awarded the Chemistry Nobel Prize for their invention of partition chromatography.[1] Since then, the technology has advanced rapidly. Researchers found that the principles underlying Tsvet's chromatography could be applied in many different ways, giving rise to the different varieties of chromatography described below. Simultaneously, advances continually improved the technical performance of chromatography, allowing the separation of increasingly similar molecules. Chromatography terms
Chromatography theoryChromatography is a physical method of separation in which the components to be separated are distributed between two phases, one which is the stationary (stationary phase) while the other (the mobile phase) moves in a definite direction. In chromatographic methods, separations results from differences in the distribution constants of the individual sample components between the two phases. In gas chromatography (GC) separations are achieved by distribution of a solute between an immobile solid or liquid stationary phase and a gas phase that percolates over the stationary phase . Sample molecules spend part of the time in the mobile phase and the other part in the stationary phase during the passage through the column. The time for an unretained solute to reach the detector from the point of injection is called the column dead time or the hold up time(tM). The solute retention time (tR) is the time difference between sample injection and the detector sensing the maximum of the retained peak. The amount of time solute molecules spend in the stationary phase is called the adjusted retention time (tR’). tR= tR’+ tM Retention factor (or capacity factor) is more fundamentally important than the absolute retention time. This represents the ratio of the time spent by solute in the stationary phase to the time it spends in the mobile phase. k= tR’/ tM = (tR - tM)/ tM In gas chromatography it is usually more convenient to measure the retention factor, k, than the gas-liquid partition coefficient, K, which requires exact knowledge of the column phase ratio. The gas-liquid partition coefficient is the relevant free energy parameter for modeling retention but this is of no great consequence since the partition coefficient is related to the retention factor by the relationship K = bk where b is the column phase ratio (volume of gas phase/volume of stationary phase).
Solvation Parameter modelAbraham’s solvation parameter model is a powerful tool to characterize the retention properties of open-tubular columns used in gas chromatography. The general purpose of these studies was to establish quantitative relationships between stationary phase composition and chromatographic selectivity, to identify selectivity equivalent stationary phases of different composition, and to assess the effect of temperature on chromatographic selectivity. According to solvation parameter model for the transfer of a solute from a gas phase to a condensed phase, Equation is given below: Log SP=c + eE +sS + aA + bB + lL SP is some free energy related solute property such as a distribution constant, retention factor, specific retention volume, relative adjusted retention time, or retention index value. The lower case letters (e, s, a, b, and l) are system constants representing the contribution of the stationary phase to retention through its ability to interact with a solute by defined intermolecular interactions. The capital letters (E, S, A, B, and L) are solute descriptors for complimentary interactions with the system constants of the stationary phase. The model intercept, is not usually assigned any physical significance, and is used only to calculate retention on individual columns. It is dominated by the phase ratio when the dependent variable is the retention factor. The system constants are identified as the opposing contributions of cavity formation and dispersion interactions, l, the contribution from interactions with lone pair electrons, e, the contribution from dipole-type interactions, s, the contribution from hydrogen-bond basicity of the stationary phase (because a basic phase will interact with an acid solute), a, and b the contribution from the hydrogen-bond acidity of the stationary phase. The system constants and their variation with temperature are used to characterize stationary phase selectivity and the effect of temperature on selectivity. Plate theoryThe plate theory of chromatography was developed by Archer John Porter Martin and Richard Laurence Millington Synge. The plate theory describes the chromatography system, the mobile and stationary phases, as being in equilibrium. The partition coefficient K is based on this equilibrium, and is defined by the following equation: K is assumed to be independent of concentration, and can change if experimental conditions are changed, for example temperature is increased or decreased. As K increases, it takes longer for solutes to separate. For a column of fixed length and flow, the retention time (tR) and retention volume (Vr) can be measured and used to calculate K. Techniques by chromatographic bed shapeColumn chromatography
Column chromatography is a separation technique in which the stationary bed is within a tube. The particles of the solid stationary phase or the support coated with a liquid stationary phase may fill the whole inside volume of the tube (packed column) or be concentrated on or along the inside tube wall leaving an open, unrestricted path for the mobile phase in the middle part of the tube (open tubular column). Differences in rates of movement through the medium are calculated to different retention times of the sample.[2] In 1978, W. C. Still introduced a modified version of column chromatography called flash column chromatography (flash).[3] The technique is very similar to the traditional column chromatography, except for that the solvent is driven through the column by applying positive pressure. This allowed most separations to be performed in less than 20 minutes, with improved separations compared to the old method. Modern flash chromatography systems are sold as pre-packed plastic cartridges, and the solvent is pumped through the cartridge. Systems may also be linked with detectors and fraction collectors providing automation. The introduction of gradient pumps resulted in quicker separations and less solvent usage. In expanded bed adsorption, a fluidized bed is used, rather than a solid phase made by a packed bed. This allows omission of initial clearing steps such as centrifugation and filtration, for culture broths or slurries of broken cells. Planar Chromatography
Planar chromatography is a separation technique in which the stationary phase is present as or on a plane. The plane can be a paper, serving as such or impregnated by a substance as the stationary bed (paper chromatography) or a layer of solid particles spread on a support such as a glass plate (thin layer chromatography). Paper Chromatography
Paper chromatography is a technique that involves placing a small dot of sample solution onto a strip of chromatography paper. The paper is placed in a jar containing a shallow layer of solvent and sealed. As the solvent rises through the paper it meets the sample mixture which starts to travel up the paper with the solvent. Different compounds in the sample mixture travel different distances according to how strongly they interact with the paper. This allows the calculation of an Rf value and can be compared to standard compounds to aid in the identification of an unknown substance. Thin layer chromatography
Thin layer chromatography (TLC) is a widely-employed laboratory technique and is similar to paper chromatography. However, instead of using a stationary phase of paper, it involves a stationary phase of a thin layer of adsorbent like silica gel, alumina, or cellulose on a flat, inert substrate. Compared to paper, it has the advantage of faster runs, better separations, and the choice between different adsorbents. Different compounds in the sample mixture travel different distances according to how strongly they interact with the adsorbent. This allows the calculation of an Rf value and can be compared to standard compounds to aid in the identification of an unknown substance. Techniques by physical state of mobile phaseGas chromatography
Gas chromatography (GC), also sometimes known as Gas-Liquid chromatography, (GLC), is a separation technique in which the mobile phase is a gas. Gas chromatography is always carried out in a column, which is typically "packed" or "capillary" (see below) . Gas chromatography (GC) is based on a partition equilibrium of analyte between a solid stationary phase (often a liquid silicone-based material) and a mobile gas (most often Helium). The stationary phase is adhered to the inside of a small-diameter glass tube (a capillary column) or a solid matrix inside a larger metal tube (a packed column). It is widely used in analytical chemistry; though the high temperatures used in GC make it unsuitable for high molecular weight biopolymers or proteins (heat will denature them), frequently encountered in biochemistry, it is well suited for use in the petrochemical, environmental monitoring, and industrial chemical fields. It is also used extensively in chemistry research. Liquid chromatographyLiquid chromatography (LC) is a separation technique in which the mobile phase is a liquid. Liquid chromatography can be carried out either in a column or a plane. Present day liquid chromatography that generally utilizes very small packing particles and a relatively high pressure is referred to as high performance liquid chromatography (HPLC). In the HPLC technique, the sample is forced through a column that is packed with irregularly or spherically shaped particles or a porous monolithic layer (stationary phase) by a liquid (mobile phase) at high pressure. HPLC is historically divided into two different sub-classes based on the polarity of the mobile and stationary phases. Technique in which the stationary phase is more polar than the mobile phase (e.g. toluene as the mobile phase, silica as the stationary phase) is called normal phase liquid chromatography (NPLC) and the opposite (e.g. water-methanol mixture as the mobile phase and C18 = octadecylsilyl as the stationary phase) is called reversed phase liquid chromatography (RPLC). Ironically the "normal phase" has fewer applications and RPLC is therefore used considerably more. Specific techniques which come under this broad heading are listed below. It should also be noted that the following techniques can also be considered fast protein liquid chromatography if no pressure is used to drive the mobile phase through the stationary phase. See also Aqueous Normal Phase Chromatography. Affinity chromatography
Affinity chromatography is based on selective non-covalent interaction between an analyte and specific molecules. It is very specific, but not very robust. It is often used in biochemistry in the purification of proteins bound to tags. These fusion proteins are labelled with compounds such as His-tags, biotin or antigens, which bind to the stationary phase specifically. After purification, some of these tags are usually removed and the pure protein is obtained. Supercritical fluid chromatography
Supercritical fluid chromatography is a separation technique in which the mobile phase is a fluid above and relatively close to its critical temperature and pressure. Techniques by separation mechanismIon exchange chromatography
Ion exchange chromatography utilizes ion exchange mechanism to separate analytes. It is usually performed in columns but the mechanism can be benefited also in planar mode. Ion exchange chromatography uses a charged stationary phase to separate charged compounds including amino acids, peptides, and proteins. In conventional methods the stationary phase is an ion exchange resin that carries charged functional groups which interact with oppositely charged groups of the compound to be retained. Ion exchange chromatography is commonly used to purify proteins using FPLC. Size exclusion chromatography
Size exclusion chromatography (SEC)is also known as gel permeation chromatography (GPC)or gel filtration chromatography and separates molecules according to their size (or more accurately according to their hydrodynamic diameter or hydrodynamic volume). Smaller molecules are able to enter the pores of the media and; therefore, take longer to elute, whereas larger molecules are excluded from the pores and elute faster. It is generally a low resolution chromatography technique and thus it is often reserved for the final, "polishing" step of a purification. It is also useful for determining the tertiary structure and quaternary structure of purified proteins, especially since it can be carried out under native solution conditions. Special techniquesReversed-phase chromatography
Reversed-phase chromatography is an elution procedure used in liquid chromatography in which the mobile phase is significantly more polar than the stationary phase. Two-dimensional chromatographyIn some cases, the chemistry within a given column can be insufficient to separate some analytes. It is possible to direct a series of unresolved peaks onto a second column with different physico-chemical (Chemical classification) properties. Since the mechanism of retention on this new solid support is different from the first dimensional separation, it can be possible to separate compounds that are indistinguishable by one-dimensional chromatography. Pyrolysis gas chromatographyFast protein liquid chromatography
Fast protein liquid chromatography (FPLC) is a term applied to several chromatography techniques which are used to purify proteins. Many of these techniques are identical to those carried out under high performance liquid chromatography. Countercurrent chromatography
Countercurrent chromatography (CCC) is a type of liquid-liquid chromatography, where both the stationary and mobile phases are liquids. It involves mixing a solution of liquids, allowing them to settle into layers and then separating the layers. Chiral chromatographyChiral Chromatography involves the separation of stereoisomers. In the case of enantiomers, these have no chemical or physical differences apart from being three dimensional mirror images. Conventional chromatography or other separation processes are incapable of separating them. To enable chiral separations to take place, either the mobile phase or the stationary phase must themselves be made chiral, giving differing affinities between the analytes. Chiral chromatography HPLC columns (with a chiral stationary phase) in both normal and reversed phase are commercially available. See also
References
Categories: Separation processes | Chemical pathology | Chromatography |
|||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Chromatography". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||