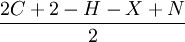To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Degree of unsaturationThe degree of unsaturation (also known as the index of hydrogen deficiency (IHD) or rings plus double bonds[1]) formula is used in organic chemistry to help draw chemical structures. The formula lets the user determine how many rings, double bonds, and triple bonds are present in the compound to be drawn. It does not give the exact number of rings or double or triple bonds, but rather the sum of the number of rings and double bonds plus twice the number of triple bonds. The final structure is verified with use of NMR, mass and IR spectroscopy, as well as inspection. The formula for degree of unsaturation is Product highlightwhere C is the numbers of carbon atoms, H is the numbers of hydrogen atoms, X is the numbers of halide atoms, and N is the numbers of nitrogen atoms. Do not count group VI elements such as oxygen and sulfur atoms (but count H groups attached to oxygen atoms, such as hydroxyl groups). Halogen atoms (F, Cl, Br, I) count as H atoms for degree of unsaturation calculations. The degree of unsaturation is used to calculate the number of rings and pi bonds, where
See alsoReferences
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Degree_of_unsaturation". A list of authors is available in Wikipedia. |