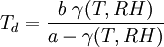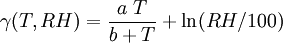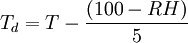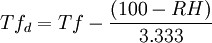To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Dew pointThe dew point (or dewpoint) is the temperature to which a given parcel of air must be cooled, at constant barometric pressure, for water vapor to condense into water. The condensed water is called dew. The dew point is a saturation point. When the dew point temperature falls below freezing it is called the frost point, as the water vapor no longer creates dew but instead creates frost or hoarfrost by deposition. The dew point is associated with relative humidity. A high relative humidity indicates that the dew point is closer to the current air temperature. If the relative humidity is 100%, the dew point is equal to the current temperature. Given a constant dew point, an increase in temperature will lead to a decrease in relative humidity. It is for this reason that equatorial climates can have low relative humidity, yet still feel humid.[citation needed] At a given barometric pressure, independent of temperature, the dew point indicates the mole fraction of water vapor in the air, and therefore determines the specific humidity of the air. The dew point is an important statistic for general aviation pilots, as it is used to calculate the likelihood of carburetor icing and fog. Additional recommended knowledge
ExplanationThe graph above shows the maximum percentage of water vapor that can exist in air at sea level across a range of temperatures. With higher temperatures, the equilibrium partial pressure of water vapor increases and more water evaporates. The behavior of water vapor does not depend on the presence of air. The formation of dew would occur at the dew point even if the only gas present were water vapor. Dew point is a monotonic function of the partial pressure of water vapor, so dew point can be determined from partial pressure of water vapor alone, and vice versa. Constant pressureAt a given barometric pressure, independent of temperature, the dew point indicates the mole fraction of water vapor in the air, or, put differently, determines the specific humidity of the air. If the barometric pressure rises without changing this mole fraction, the dew point will rise accordingly. Reducing the mole fraction will bring the dew point back down to its initial value. In the same way, increasing the mole fraction after a pressure drop brings the dew point back up to its initial level. For this reason, the same dew point in New York and Denver (which is at a much higher altitude) will imply that a higher fraction of the air in Denver consists of water vapor than in New York. Varying pressureAt a given temperature but independent of barometric pressure, the dew point indicates the absolute humidity of the air. If the temperature rises without changing the absolute humidity, the dew point will rise accordingly. Reducing the absolute humidity will bring the dew point back down to its initial value. In the same way, increasing the absolute humidity after a temperature drop brings the dew point back up to its initial level. Coming back to the New York - Denver example, this means that if the dew point and temperature in both cities are the same, then the mass of water vapor per cubic meter of air will also be the same in those cities. Human reaction to high dew pointsHumans tend to react with discomfort to high dew points, as a high dew point corresponds with a high ambient temperature (causing the body to perspire and produce sweat) and/or a high relative humidity (which inhibits the evaporation of sweat, by which the body is cooled); as a result the body may overheat, resulting in discomfort. A lower dew point, meanwhile, corresponds to a lower ambient temperature or lower relative humidity, either of which allow the body to more effectively regulate its temperature to avoid overheating. Those accustomed to continental climates often begin to feel uncomfortable when the dew point reaches between 15 and 20 °C (59 to 68 °F). Most inhabitants of these areas will consider dew points above 21 °C (70 °F) to be oppressive.
Record high dew pointThe highest recorded dew point was 35°C (95°F), reported in Dhahran, Saudi Arabia at 3 p.m. July 9, 2003. The temperature was 42.2°C (108°F) resulting in an apparent temperature of 77.7°C (172°F).[2] Calculating the dew pointA well-known approximation used to calculate the dew point Td given the relative humidty RH and the actual temperature T of air is: where where the temperatures are in degrees Celsius and "ln" refers to the natural logarithm. The constants are:
This expression is based on the "Magnus" (or "Magnus-Tetens") approximation for the saturation vapor pressure of water in air as a function of temperature.[3] It is considered valid for
Simple approximationThere is also a very simple approximation which allows conversion between the dew point, the dry bulb temperature and the relative humidity, which is accurate to within about ±1 °C as long as the relative humidity is above 50%. The equation is: or
This can be expressed as a simple rule of thumb: For every 1 °C difference in the dew point and dry bulb temperatures, the relative humidity decreases by 5%, starting with RH=100% when the dew point equals the dry bulb temperature. where in this case RH is in percent, and T and Td are in degrees Celsius. The derivation of this, a discussion of its accuracy, comparisons to other approximations, and more information on the history and applications of the dew point are given in the Bulletin of the American Meteorological Society [4]. In Fahrenheit For example, a relative humidity of 100% means dew point is same as air temp. For 90% RH dew point is 3 degrees Fahrenheit lower than air temp. For every 10 percent lower, dew point drops 3 °F. Tfd is in degrees Fahrenheit; RH same as above. See also
References
Categories: Psychrometrics | Atmospheric thermodynamics |
|||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Dew_point". A list of authors is available in Wikipedia. |