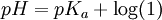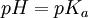To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Titration
Titration is a common laboratory method of quantitative/chemical analysis that can be used to determine the concentration of a known reactant. Because volume measurements play a key role in titration, it is also known as volumetric analysis. A reagent, called the titrant, of known concentration (a standard solution) and volume is used to react with a solution of the analyte, whose concentration is not known in advance. Using a calibrated burette to add the titrant, it is possible to determine the exact amount that has been consumed when the endpoint is reached. The endpoint is the point at which the titration is complete, as determined by an indicator (see below). This is ideally the same volume as the equivalence point - the volume of added titrant at which the number of moles of titrant is equal to the number of moles of analyte, or some multiple thereof (as in polyprotic acids). In the classic strong acid-strong base titration, the endpoint of a titration is the point at which the pH of the reactant is just about equal to 7, and often when the solution permanently changes color due to an indicator. There are however many different types of titrations (see below). Many methods can be used to indicate the endpoint of a reaction; titrations often use visual indicators (the reactant mixture changes colour). In simple acid-base titrations a pH indicator may be used, such as phenolphthalein, which becomes pink when a certain pH (about 8.2) is reached or exceeded. Another example is methyl orange, which is red in acids and yellow in alkali solutions. Not every titration requires an indicator. In some cases, either the reactants or the products are strongly coloured and can serve as the "indicator". For example, an oxidation-reduction titration using potassium permanganate (pink/purple) as the titrant does not require an indicator. When the titrant is reduced, it turns colourless. After the equivalence point, there is excess titrant present. The equivalence point is identified from the first faint pink colour that persists in the solution being titrated. Due to the logarithmic nature of the pH curve, the transitions are, in general, extremely sharp; and, thus, a single drop of titrant just before the endpoint can change the pH significantly — leading to an immediate colour change in the indicator. There is a slight difference between the change in indicator color and the actual equivalence point of the titration. This error is referred to as an indicator error, and it is indeterminate. Additional recommended knowledge
History and etymologyThe word "titration" comes from the Latin word titalus, meaning inscription or title. The French word titre, also from this origin, means rank. Titration, by definition, is the determination of rank or concentration of a solution with respect to water with a pH of 7 (which is the pH of pure water). The origins of volumetric analysis are in late-18th-century French chemistry. Francois Antoine Henri Descroizilles developed the first burette (which looked more like a graduated cylinder) in 1791. Joseph Louis Gay-Lussac, this gay developed an improved version of the burette that included a side arm, and coined the terms "pipette" and "burette" in an 1824 paper on the standardization of indigo solutions. A major breakthrough in the methodology and popularization of volumetric analysis was due to Karl Friedrich Mohr, who redesigned the burette by placing a clamp and a tip at the bottom, and wrote the first textbook on the topic, Lehrbuch der chemisch-analytischen Titrirmethode (Textbook of analytical-chemical titration methods), published in 1855.[1] Preparing a sample for titrationIn a titration, both titrant and analyte are required to be aqueous, or in a solution form. If the sample is not a liquid or solution, the samples must be dissolved. If the analyte is very concentrated in the sample, it might be useful to dilute the sample. Although the vast majority of titrations are carried out in aqueous solution, other solvents such as glacial acetic acid or ethanol (in petrochemistry) are used for special purposes. A measured amount of the sample can be given in the flask and then be dissolved or diluted. The mathematical result of the titration can be calculated directly with the measured amount. Sometimes the sample is dissolved or diluted beforehand, and a measured amount of the solution is used for titration. In this case the dissolving or diluting must be done accurately with a known coefficient because the mathematical result of the titration must be multiplied with this factor. Many titrations require buffering to maintain a certain pH for the reaction. Therefore, buffer solutions are added to the reactant solution in the flask. Some titrations require "masking" of a certain ion. This can be necessary when two reactants in the sample would react with the titrant and only one of them must be analysed, or when the reaction would be disturbed or inhibited by this ion. In this case another solution is added to the sample, which "masks" the unwanted ion (for instance by a weak binding with it or even forming a solid insoluble substance with it). Some redox reactions may require heating the solution with the sample and titration while the solution is still hot (to increase the reaction rate). ProcedureA typical titration begins with a beaker or Erlenmeyer flask containing a precise volume of the reactant and a small amount of indicator, placed underneath a burette containing the reagent. By controlling the amount of reagent added to the reactant, it is possible to detect the point at which the indicator changes colour. As long as the indicator has been chosen correctly, this should also be the point where the reactant and reagent neutralise each other, and, by reading the scale on the burette, the volume of reagent can be measured. As the concentration of the reagent is known, the number of moles of reagent can be calculated (since concentration = moles / volume). Then, from the chemical equation involving the two substances, the number of moles present in the reactant can be found. Finally, by dividing the number of moles of reactant by its volume, the concentration is calculated. Titration curves
In monoprotic acids, the point halfway between the beginning of the curve (before any titrant has been added) and the equivalence point is significant: at that point, the concentrations of the two solutions (the titrant and the original solution) are equal. Therefore, the Henderson-Hasselbalch equation can be solved in this manner: Therefore, one can easily find the acid dissociation constant of the monoprotic acid by finding the pH of the point halfway between the beginning of the curve and the equivalence point, and solving the simplified equation. In the case of the sample curve, the Ka would be approximately 1.78×10-5 from visual inspection (the actual Ka2 is 1.7×10-5) For polyprotic acids, calculating the acid dissociation constants is only marginally more difficult: the first acid dissociation constant can be calculated the same way as it would be calculated in a monoprotic acid. The second acid dissociation constant, however, is the point halfway between the first equivalence point and the second equivalence point (and so on for acids that release more than two protons, such as phosphoric acid). Types of titrationsTitrations can be classified by the type of reaction. Different types of titration reaction include:
Measuring the endpoint of a titrationDifferent methods to determine the endpoint include:
Other termsThe term back titration is used when a titration is done "backwards": instead of titrating the original analyte, one adds a known excess of a standard reagent to the solution, then titrates the excess. A back titration is useful if the endpoint of the reverse titration is easier to identify than the endpoint of the normal titration. They are also useful if the reaction between the analyte and the titrant is very slow. Particular uses
References
|
|||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Titration". A list of authors is available in Wikipedia. | |||||||||||||||





![pH = pK_a + \log \left( \frac{[\mbox{base}]}{[\mbox{acid}]} \right)](images/math/5/d/9/5d91d139a60e7c7ec84dbdb2d7aeec0a.png)