To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Troposphere
The troposphere is the lowest portion of Earth's atmosphere. It contains approximately 75% of the atmosphere's mass and almost all of its water vapor and aerosols. The average depth of the troposphere is about 11 km in the middle latitudes. It is deeper in the tropical regions (up to 20 km) and shallower near the poles (about 7 km in summer, indistinct in winter). The lowest part of the troposphere, where friction with the Earth's surface influences air flow, is the planetary boundary layer. This layer is typically a few hundred meters to 2 km deep depending on the landform and time of day. The border between the troposphere and stratosphere, called the tropopause, is a temperature inversion.[1] The word troposphere derives from the Greek "tropos" for "turning" or "mixing," reflecting the fact that turbulent mixing plays an important role in the troposphere's structure and behavior. Most of the phenomena we associate with day-to-day weather occur in the troposphere.[1] Additional recommended knowledge
Pressure and temperature structureCompositionThe chemical composition of the troposphere is essentially uniform, with the notable exception of water vapor. The source of water vapor is at the surface through the processes of evaporation and transpiration. Furthermore the temperature of the troposphere decreases with height, and saturation vapor pressure decreases strongly with temperature, so the amount of water vapor that can exist in the atmosphere decreases strongly with height. Thus the proportion of water vapor is normally greatest near the surface and decreases with height. PressureThe pressure of the atmosphere is maximum at the surface and decreases with higher altitude. This is because the atmosphere is very nearly in hydrostatic equilibrium, so that the pressure is equal to the weight of air above a given point. The change in pressure with height therefore can be equated to the density with this hydrostatic equation:[2] where:
Since temperature in principle also depends on altitude, one needs a second equation to determine the pressure as a function of height, as discussed in the next section. TemperatureThe temperature of the troposphere generally decreases with altitude. The rate at which the temperature decreases, dT / dz, is called the lapse rate. The reason for this decrease is as follows. When the air is stirred by convection, and a parcel of air rises, it expands, because the pressure is lower at higher altitudes. As the air parcel expands, it pushes on the air around it, doing work; but generally it does not gain heat in exchange from its environment, because its thermal conductivity is low (such a process is called adiabatic). Since the parcel does work and gains no heat, it loses energy, and so its temperature decreases. (The reverse, of course, will be true for a sinking parcel of air.) [1] Since the heat exchanged dQ is related to the entropy change dS by dQ=T dS, the equation governing the temperature as a function of height for a thoroughly mixed atmosphere is where S is the entropy. The rate at which temperature decreases with height under such conditions is called the adiabatic lapse rate. For dry air, which is approximately an ideal gas, we can proceed further. The adiabatic equation for an ideal gas is [3] where γ is the heat capacity ratio (γ=7/5, for air). Combining with the equation for the pressure, one arrives at the dry adiabatic lapse rate,[4] If the air contains water vapor, then cooling of the air can cause the water to condense, and the behavior is no longer that of an ideal gas. If the air is at the saturated vapor pressure, then the rate at which temperature drops with height is called the saturated adiabatic lapse rate. More generally, the actual rate at which the temperature drops with altitude is called the environmental lapse rate. In the troposphere, the average environmental lapse rate is a drop of about 6.5 °C for every 1 km (1000 meters) increase in height. [1] Depending on the weather conditions, one may find that the environmental lapse rate (the actual rate at which temperature drops with height, dT / dz) is not equal to the adiabatic lapse rate (or correspondingly, that Temperatures decrease at middle latitudes from an average of 15°C at sea level to about -55°C at the beginning of the tropopause. At the poles, the troposphere is thinner and the temperature only decreases to -45°C, while at the equator the temperature at the top of the troposphere can reach -75°C.[citation needed] TropopauseThe tropopause is the boundary region between the troposphere and the stratosphere. Measuring the temperature change with height through the troposphere and the stratosphere identifies the location of the tropopause. In the troposphere, temperature decreases with altitude. In the stratosphere, however, the temperature remains constant for a while and then increases with altitude. The region of the atmosphere where the lapse rate changes from positive (in the troposphere) to negative (in the stratosphere), is defined as the tropopause.[1] Thus, the tropopause is an inversion layer, and there is little mixing between the two layers of the atmosphere. Atmospheric circulationThe basic structure of large-scale circulation in the troposphere remains fairly constant. There are three convection cells in each hemisphere: the Hadley cell, the Ferrel cell, and the Polar cell, which guide the prevailing winds, thereby transporting heat from the equator to the poles.[1] See also
References
|
|||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Troposphere". A list of authors is available in Wikipedia. |





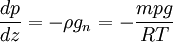
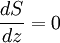
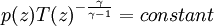
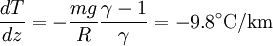
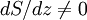 ). If the upper air is warmer than predicted by the adiabatic lapse rate (
). If the upper air is warmer than predicted by the adiabatic lapse rate (

