Lone water molecules turn out to be directors of supramolecular chemistry
Scientists in supramolecular chemistry often run into surprising outcomes. A broken seal of a lab cuvette led an American researcher at the Eindhoven University of Technology to the origin of these inexplicable results: the weather. Or the humidity, to be more precise, because this determines the water concentration in oils used as solvents, which was previously thought to be negligible. Research now shows that the lone water molecules in oil aren't just spectators, they firmly direct supramolecular processes. This outcome means that a lot of previous research has to be re-examined, but also that chemists get a new, cheap and powerful tool.
In September 2016 postdoctoral researcher Nathan van Zee spent a week with his parents in Florida. But his thoughts often led him back to his lab in The Netherlands because he had been struggling for quite some time with erratic outcomes he didn't understand. The molecular helices that he was synthesizing sometimes had a clockwise structure and sometimes an anti-clockwise structure. This is an important difference--for instance, it can make the difference between a drug that works and a drug that doesn't. Van Zee had already tried all sorts of things, but was not getting any closer to answers.
After his flight back from the US he immediately went to his laboratory to start a new experiment, after which he wanted to take a nap. But his jetlag caused him to use the wrong settings for the instrument, and he overslept. His eureka moment, which would eventually lead to a Nature publication, came the next day. Back in the lab he saw strange results. But he also saw that the seal of his lab cuvette was broken, and the ultradry air of the enclosing sample holder had gotten into it. This gave him an important clue: his sample had become drier, suggesting the water content of the solvent oil is a driving force.
Van Zee and his supervisor professor Bert Meijer immediately knew they were on to something important, and they decided to get to the bottom of it. They discovered that it was indeed the water concentration in the sample that made the difference, even though it was only a few ppm (parts per million). Even with extremely small fluctuations in water concentration, they observed that the helix rotation changes from clockwise to anti-clockwise.
This result caused the researchers to have a closer look at some of their previous work that had inexplicable results. It turned out that also in these tests the water concentration determined the outcome. The previously inexplicable changes in the results were caused by fluctuations in the water content of the oil-based solvent. Because that content fluctuates with the humidity. And the atmospheric humidity--indoors and outdoors--is constantly changing because of the weather. The consequence is that a test run on day A can have totally different results from exactly the same test run on day B.
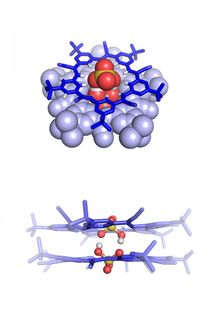
Van Zee and his colleagues also revealed how the minute water concentration can have such a big impact. Water molecules are polar: one side is negatively charged and the other positively. That's why they like to bond, via so-called hydrogen bonds. But oil is hydrophobic: it repels water. This repulsion leaves too little space for the water molecules in oil to bond with other water molecules; they are isolated. That means their potential energy to form new hydrogen bonds is available for other uses. The Eindhoven researchers have shown that this energy of water plays a crucial role in the formation of supramolecular structures. These are molecular aggregates based on reversible bonds, for example hydrogen bonds.
Their findings put quite a burden on their own science field as well as adjacent ones. A lot of chemistry is done in oil, so a lot of previous research will have to be re-evaluated to assess the effect of water. The researchers suspect that many previous reports of unexplained phenomena, be it changes in structure, size or processing, are fundamentally due to interactions with water.
On the other hand the Eindhoven research team provides chemists worldwide with an amazing new instrument, Van Zee explains. "The water concentration turns out to have a very strong influence. And one can easily control it, by manipulating the humidity, which is pretty simple in a closed environment. We think there will be a lot of follow-up research also in other fields of chemistry."