Description of rotating molecules made easy
New numerical technique to describe molecules in solvents
Feynman diagrams are a powerful tool in condensed matter physics. By turning highly complex equations into sets of simple diagrams, the method has established itself as one of the sharpest tools in a theoretical physicist’s toolbox. Giacomo Bighin, a postdoc in the group of Mikhail Lemeshko at the Institute of Science and Technology Austria (IST Austria), has now extended the Feynman diagram technique: originally devised for subatomic particles, the simplest objects imaginable, the technique can now work with molecules, far more complex objects. The research, which was published in the journal “Physical Review Letters”, is expected to drastically simplify the description of molecular rotations in solvents. This brings scientists one step closer to their long-term goal of understanding chemical reactions in solvents at the microscopic level and, potentially, controlling them.
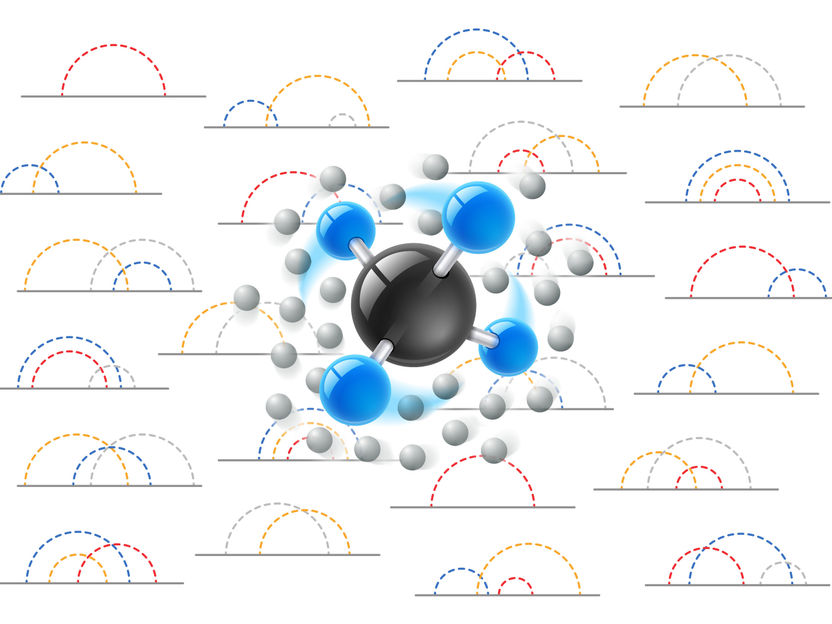
Feynman diagrams can now be used to describe rotating molecules in solvents.
IST Austria/Birgit Rieger
Sometimes when you’re stuck on a problem, the solution could be closer than you think, for instance in a different area of the research field you are working in. But thinking across disciplines is difficult and requires a good mix of expertise and an environment that fosters such interdisciplinary collaborations. Giacomo Bighin found such an environment at IST Austria when he, a condensed matter physicist, joined the group of Mikhail Lemeshko, a molecular physicist. The result is a new method for molecular physics, one that can greatly facilitate the description of rotating molecules in solvents and paves the way for eventually controlling their reactions.
“Molecules always rotate, and how they interact with one another depends on their relative orientation. That is, if they hit another molecule with one end, it has a different effect than if they hit it with the other end,” explains Mikhail Lemeshko. The orientation of molecules and hence chemical reactions have already been controlled in experiments on molecular gases, but it is quite challenging to do the same in solvents. This is a long-term goal that Mikhail Lemeshko and his group are working towards, one step at a time. The step they have just taken is about being better able to describe the rotation of a molecule in a solvent—a prerequisite for eventually controlling reactions in this environment.
Transferring the method, however, was not easy. “Feynman diagrams work for point-like particles such electrons. Point-like means that they are not affected by rotations: if you rotate an electron, it looks exactly the same as before. Molecules, on the other hand, are more complex and can rotate and change their orientation in space” explains Giacomo Bighin. In order to transfer the method from electrons to molecules, he had to develop a new formalism. Previously, it was not known if it would even work for molecules, and adapting the method took Bighin more than a year. Now the formalism is ready to use in chemical problems. “We expect that people from a more molecular background will see that it is now possible to study molecules in this way. The technique delivers extremely precise results in condensed matter physics, and it has the potential to achieve the same accuracy in molecular simulations,” Lemeshko adds.
Original publication
Other news from the department science

Get the chemical industry in your inbox
From now on, don't miss a thing: Our newsletter for the chemical industry, analytics, lab technology and process engineering brings you up to date every Tuesday and Thursday. The latest industry news, product highlights and innovations - compact and easy to understand in your inbox. Researched by us so you don't have to.