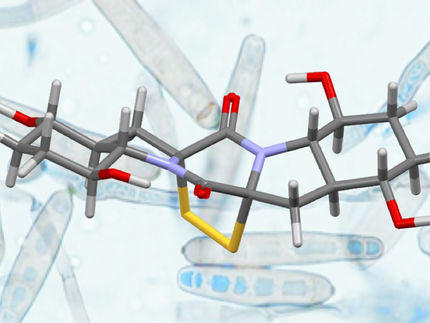
The first carbene transfer reaction between main group element compounds
Advertisement
Scientists in Germany have made a new, versatile and inexpensive carbene transfer reagent that can be used to make both main group and transition metal carbene complexes. It is the first example of a carbene transfer reaction between main group element compounds.
N-heterocyclic carbene (NHC) complexes are commonly used in catalysis but the synthesis and isolation of carbene ligands is an ongoing challenge. Researchers have only seen any real success using NHCs with bulky substituents at the N,N’-positions, as they provide steric protection to the carbene centre and so make the NHC more stable.
Now, the team has developed a new NHC-transfer reagent based on SiCl4 ((NHCMe)SiCl4 (NHCMe = 1,3-dimethylimidazolidin-2-ylidene). They made it from silicochloroform, which is easily accessible as it is a raw material in the chemical industry.
They demonstrated the reagent’s utility by transferring the sterically non-demanding NHC-ligand to the phosphorus(III). Previous attempts to make sterically non-demanding carbene complexes of phosphorus(III) only led to decomposition and/or the formation of cationic species.
The experimental results are supported by DFT calculations, which concluded a similar carbene transfer potential compared to established transfer reagents based on silver(I) chloride. The new reagent is therefore a low-cost alternative to the established yet expensive sliver-based carbene transfer reagents.
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.