Nano-level lubricant tuning improves material for electronic devices and surface coatings
molybdenum disulfide (MoS2), which is ubiquitously used as a solid lubricant, has recently been shown to have a two-dimensional (2D) form that is similar to graphene. But, when thinned down to less than a nanometer thick, MoS2 demonstrates properties with great promise as a functional material for electronic devices and surface coatings.

This is a scanning electron microscope image of atomically-thin MoS2 with hierarchical, dual-scale structures.
SungWoo Nam, University of Illinois
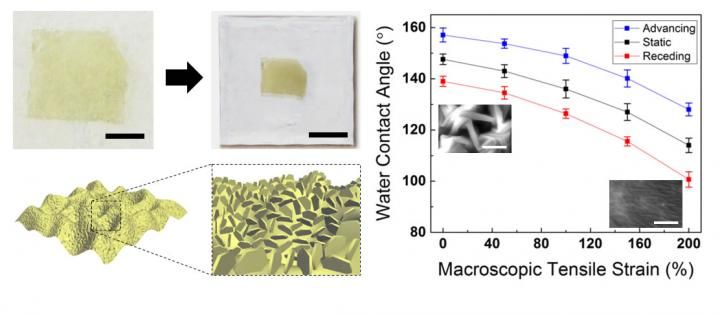
Image shows how controlled and dynamic surface architecturing/roughening of atomically thin MoS2 affects wettability.
SungWoo Nam, University of Illinois

Images of atomically thin MoS2 with micro- and nano-scale roughnesses and their corresponding water contact angles.
SungWoo Nam, University of Illinois



Researchers at the University of Illinois at Urbana-Champaign have developed a new approach to dynamically tune the micro- and nano-scale roughness of atomically thin MoS2, and consequently the appropriate degree of hydrophobicity for various potential MoS2-based applications.
"The knowledge of how new materials interact with water is a fundamental," explained SungWoo Nam, an assistant professor of mechanical science and engineering at Illinois. "Whereas the wettability of its more famous cousin, graphene, has been substantially investigated, that of atomically thin MoS2--in particular atomically thin MoS2 with micro- and nano-scale roughness--has remained relatively unexplored despite its strong potential for fundamental research and device applications. Notably, systematic study of how hierarchical microscale and nanoscale roughness of MoS2 influence its wettability has been lacking in the scientific community."
"This work will provide a new approach to dynamically tune the micro- and nano-scale roughness of atomically thin MoS2 and consequently the appropriate degree of hydrophobicity for various potential MoS2-based applications," stated Jonghyun Choi, a mechanical engineering graduate student and first author of the article. "These include waterproof electronic devices with superhydrophobicity with water contact angle greater than 150 degrees. It may also be useful for medical applications with reduced hydrophobicity (WCA less than 100 degrees) for effective contact with biological substances. "
According to the authors, this study, expands the toolkit to allow tunable wettability of 2D materials, many of which are just beginning to be discovered.
"When deformed and patterned to produce micro- and nano-scale structures, MoS2 shows promise as a functional material for hydrogen evolution catalysis systems, electrodes for alkali metal-ion batteries, and field-emission arrays," Nam added. "The results should also contribute to future MoS2-based applications, such as tunable wettability coatings for desalination and hydrogen evolution."
Original publication
Other news from the department science

Get the chemical industry in your inbox
From now on, don't miss a thing: Our newsletter for the chemical industry, analytics, lab technology and process engineering brings you up to date every Tuesday and Thursday. The latest industry news, product highlights and innovations - compact and easy to understand in your inbox. Researched by us so you don't have to.