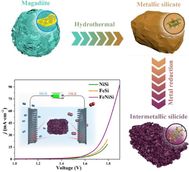
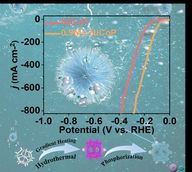
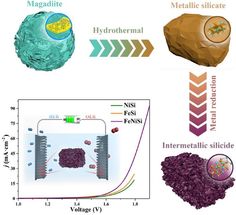
Water splitting - 84 News
Tip: You can further refine your news selection by departments, organizations, countries and industries.

Water splitting - News by department
Loading...
Water splitting - News by industry
Water splitting - News by organisation
Water splitting - News by country
Loading...
Haven't found the right news yet?
The chemeurope.com news search
Start your targeted search now with a wide range of filter options. Our database includes an archive of 25,629 chemistry news from business and science, which you can search by topic and organization, filtered by time and geography.