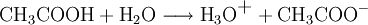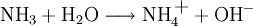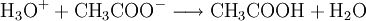To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Brønsted-Lowry acid-base theoryIn chemistry, the Bronsted-Lowry acid-base theory or Bronsted-Lowry concept is an acid-base theory describing the reaction mechanism between acids and bases, and was independently proposed by Johannes Nicolaus Bronsted and Thomas Martin Lowry in 1923. In this system, a Bronsted-Lowry acid is defined as any species which has the tendency to lose, or "donate" a hydrogen ion (proton), and a Brønsted-Lowry base as a substance with a tendency to gain or "accept" a hydrogen ion (proton). Thus, other substances such as water molecules can have behaviours not defined in the common definition of an acid or base. Additional recommended knowledgeIn the reaction between acetic acid and water, acetic acid acts as an acid by donating a proton to water, which acts as a base. The equation given is: Water can also act as an acid, for instance when it reacts with ammonia. The equation given for this reaction is: in which H2O donates a proton to NH3. Thus water is amphoteric: able to act as either an acid or a base. These reactions can act in reverse as well as in the forward direction. The reverse of the reaction between acetic acid and water is: In the reverse reactions H3O+ acts as an acid to donate a proton to CH3COO-, which is acting as a base. In the forward reaction, CH3COOH acts as an acid to donate a proton, and water as a base to receive it. H2O and H3O+ are therefore considered one acid-base conjugate pair, and CH3COOH and CH3COO- as another conjugate pair. See also
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Brønsted-Lowry_acid-base_theory". A list of authors is available in Wikipedia. |