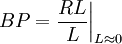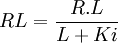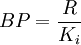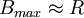To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Binding potentialIn pharmacokinetics and receptor-ligand kinetics the binding potential (BP) is a combined measure of the density of "available" neuroreceptors and the affinity of a drug to that neuroreceptor. Product highlight
DescriptionConsider a ligand receptor binding system. Ligand with a concentration L associates with a receptor of concentration or availability R to form a ligand-receptor complex with concentration RL. The binding potential is then the ratio ligand-receptor complex to free ligand at equilibrium and in the limit of L tending to 0, and is given symbol BP:
This quantity, originally defined by Mintun[1], describes the capacity of a receptor to bind ligand. It is a limit (L << Ki) of the general receptor association equation:
and is thus also equivalent to:
These equations apply equally when measuring the total receptor density or the residual receptor density available after binding to second ligand - availability. BP in Positron Emission TomographyBP is a pivotal measure in the use of positron emission tomography (PET) to measure the density of "available" receptors, e.g. to assess the occupancy by drugs or to characterize neuropsychiatric diseases (yet, one should keep in mind that binding potential is a combined measure that depends on receptor density as well as on affinity). An overview of the related methodology is e.g. given in Laruelle et al (2002)[2]. Estimating BP with PET usually requires that a reference tissue is available. A reference tissue has negligible receptor density and its distribution volume should be the same as the distribution volume in the target region if all receptors were blocked. Although the BP can be measured in a relatively unbiased way by measuring the whole time course of labelled ligand association and blood radioactivity, this is practically not always necessary. Two other common measures have been derived, which involve assumptions, but result in measures that should correlate with BP: BP1 and BP2.
Definitions and SymbolsWhile BP1 and BP2 are nonambiguous symbols, BP is not. There are many publications in which BP denotes BP2. Generally, if there were no arterial samples ("noninvasive imaging"), BP denotes BP2. Bmax: Total density of receptors = R + RL. In PET imaging, the amount of radioligand is usually very small (L << Ki, see above), thus k3 and k4: Transfer rate constants from the two tissue compartment model. See alsoReferences
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Binding_potential". A list of authors is available in Wikipedia. |