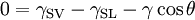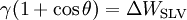To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Contact angleThe contact angle is the angle at which a liquid/vapor interface meets the solid surface. The contact angle is specific for any given system and is determined by the interactions across the three interfaces. Most often the concept is illustrated with a small liquid droplet resting on a flat horizontal solid surface. The shape of the droplet is determined by the Young-Laplace equation. The contact angle plays the role of a boundary condition. Contact angle is measured using a contact angle goniometer. The contact angle is not limited to a liquid/vapour interface; it is equally applicable to the interface of two liquids or two vapours.
Product highlight
Typical contact anglesConsider a liquid drop on a solid surface. If the liquid is very strongly attracted to the solid surface (for example water on a strongly hydrophilic solid) the droplet will completely spread out on the solid surface and the contact angle will be close to 0°. Less strongly hydrophilic solids will have a contact angle up to 90°. On many highly hydrophilic surfaces, water droplets will exhibit contact angles of 0° to 30°. If the solid surface is hydrophobic, the contact angle will be larger than 90° . On highly hydrophobic surfaces the surfaces have water contact angles as high as 150° or even nearly 180°. On these surfaces, water droplets simply rest on the surface, without actually wetting to any significant extent. These surfaces are termed superhydrophobic and can be obtained on fluorinated surfaces (Teflon-like coatings) that have been appropriately micropatterned. This is called the Lotus effect, as these new surfaces are based on lotus plants' surface (which has little protuberances) and would be superhydrophobic even to honey. The contact angle thus directly provides information on the interaction energy between the surface and the liquid. Sometimes the contact angle is measured through the gas instead of through the liquid, which reverses 0 and 180 in the above explanation. ThermodynamicsThe theoretical description of contact arises from the consideration of a thermodynamic equilibrium between the three phases: the liquid phase of the droplet (L), the solid phase of the substrate (S), and the gas/vapor phase of the ambient (V) (which will be a mixture of ambient atmosphere and an equilibrium concentration of the liquid vapor). The V phase could also be another (immiscible) liquid phase. At equilibrium, the chemical potential in the three phases should be equal. It is convenient to frame the discussion in terms of the interfacial energies. We denote the solid-vapor interfacial energy (see surface energy) as γSV, the solid-liquid interfacial energy as γSL and the liquid-vapor energy (i.e. the surface tension) as simply γ, we can write an equation that must be satisfied in equilibrium (known as the Young Equation): where θ is the experimental contact angle. Thus the contact angle can be used to determine an interfacial energy (if other interfacial energies are known). This equation can be rewritten as the Young-Dupré equation: where ΔWSLV is the adhesion energy per unit area of the solid and liquid surfaces when in the medium V.
Measuring methods
See also
References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Contact_angle". A list of authors is available in Wikipedia. |