To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Decay correct
Decay correct is a method of adjusting the measurements of radioactive decay obtained at two different time points so that they may be used as a single data set. If a chemical compound is tagged with a radioactive isotope then the amount of radiation released in a period of time is proportional to the number of atoms of the isotope; but as time passes some of the isotope decays and the isotope remaining (which is measured by the radiation) is no longer the same as the amount of compound present. Decay correction is a mathematical method of determining the amount of compound present given the amount of isotope remaining. Product highlightUseFor example, in biodistribution studies, it is common to track the movement of a compound of interest through an animal. To measure the compound it is chemically joined to a radionuclide. Thus the energy measured from the radioactive decay is related to the quantity of the compound of interest. Samples may be collected and counted at short time intervals (ex: 1 and 4 hours). The next samples (from another set of animals injected at the same starting time) may not be collected and counted until 24, 48, or 72 hours. Because isotopes for therapy and imaging often have a relatively short half life (under 10 days), it is necessary to adjust the data in order to compare all the time points. For example, the isotope copper-64, commonly used in medical research, has a half life of 12.7 hours. If you inject a large group of animals at "time zero", but measure the radioactivity in their organs at two later times, the later groups must be "decay corrected" to adjust for the decay that has occurred between the two time points. MathematicsThe formula for decay correcting is: where At is the raw, (uncorrected) count at time t, A0 is the count at time 0 (the decay corrected counts), "e" is the "inverse natural log" function, "k" is the decay constant (or "lambda"), and "t" is the elapsed time. The decay constant is ExampleThe decay correct might be used this way: a group of 20 animals is injected with a compound of interest on a Monday at 10:00 a.m. The compound is chemically joined to the isotope copper-64, which has a known half life of 12.7 hours, or 764 minutes. After one hour, the 5 animals in the "one hour" group are killed, dissected, and organs of interest are placed in sealed containers to await measurement. This is repeated for another 5 animals, at 2 hours, and again at 4 hours. At this point, (say, 4:00 p.m., Monday) all the organs collected so far are measured for radioactivity (a proxy of the distribution of the compound of interest). The next day (Tuesday), the "24 hour" group would be killed and dissected at 10:00 a.m., then measured for radioactivity, (say at 11:00 a.m.). In order to compare ALL the groups together, the data from the "24 hour" must be decay corrected: the radioactivity measured on the second day must be "adjusted" in order to allow a comparison to measurements from an earlier time, but of the same original material. In this case, "time zero" is Monday, 4:00 p.m., when the first three groups (1,2, and 4 hour animals organs) were measured. The "24 hour" group was measured at 11:00 a.m. Tuesday, which is 19 hours after the first groups. Start by calculating the decay constant "K". Substitute 12.7 (hours, the half life of copper-64) for t1 / 2 , giving
Next, multiply this value of "K" by the time elapsed between the first and second measures of radioactivity, 19 hours: (0.0546 x 19) = 1.0368. Change the sign, to make it -1.0368, then find the "inverse Ln"; in this case 0.3546. This value is in the denominator of the decay correcting fraction, so it is the same as multiplying the numerator by its inverse ( (A simple way to check if you are using the decay correct formula right is to put in the value of the half life in place of "t". After you perform the inverse Ln, the value should be very close to 0.5. When divided into the value "A" (for uncorrected counts), it effectively doubles them, which is the necessary correction after one half life has occurred.) In this case, the uncorrected values will be multiplied by 2.82, which corrects for 19 hours elapsing (between one and two half lives). If the radiation measured has dropped by half between the 4 hour sample and the 24 hour sample we might think that the concentration of compound in that organ has dropped by half; but applying the decay correct we see that the concentration is 0.5*2.82 so it has actually increased by 40% in that period. |
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Decay_correct". A list of authors is available in Wikipedia. |





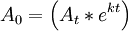
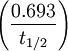 where .693 is (approximately) the
where .693 is (approximately) the 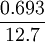 = 0.0546.
= 0.0546.
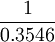 ), which is 2.82.
), which is 2.82.


