To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Dilution assayThe term dilution assay is generally used to designate a special type of bioassay in which one or more preparations (e.g. a drug) are administered to experimental units at different dose levels inducing a measurable biological response. The dose levels are prepared by dilution in a diluent that is inert in respect of the response. The experimental units can for example be cell-cultures, tissues, organs or living animals. The biological response may be quantal (e.g. positive/negative) or quantitative (e.g. growth). The goal is to relate the response to the dose, usually by interpolation techniques, and in many cases to express the potency/activity of the test preparation(s) relative to a standard of known potency/activity. Dilution assays can be direct or indirect. In a direct dilution assay the amount of dose needed to produce a specific (fixed) response is measured, so that the dose is a stochastic variable defining the tolerance distribution. Conversely, in an indirect dilution assay the dose levels are administered at fixed dose levels, so that the response is a stochastic variable. Product highlight
Statistical modelsFor a mathematical definition of a dilution assay an observation space U is defined and a function
in which e is an error term with expectation 0. F is usually assumed to be continuous and monotone. In situations where a standard preparation is included it is furthermore assumed that the test preparation T behaves like a dilution (or concentration) of the standard S
where ρ > 0 is the relative potency of T. This is the fundamental assumption of similarity of dose-response curves which is necessary for a meaningful and unambiguous definition of the relative potency. In many cases it is convenient to apply a power transformation x = zλ with λ > 0 or a logarithmic transformation x = log(z). The latter can be shown to be a limit case of
Estimates In practice, F itself is rarely of interest. More of interest is an estimate of ρ or an estimate of the dose that induces a specific response. These estimates involve taking ratios of statistically dependent parameter estimates. Fieller's theorem can be used to compute confidence intervals of these ratios. Some special cases deserve particular mention because of their widespread use: If F is linear and λ > 0 this is known as a slope-ratio model. If F is linear and λ = 0 this is known as a parallel line model. Another commonly applied model is the probit model where F is the cumulative normal distribution function, λ = 0 and e follows a binomial distribution. Example: Microbiological assay of antibiotics
SoftwareThe major statistical software packages do not cover dilution assays although a statistician should not have difficulties to write suitable scripts or macro's to that end. Several special purpose software packages for dilution assays exist. References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Dilution_assay". A list of authors is available in Wikipedia. |




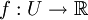 so that the responses
so that the responses 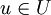 are mapped to the set of real numbers. It is now assumed that a function
are mapped to the set of real numbers. It is now assumed that a function 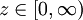 to the response
to the response
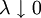 so if
so if 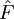 of
of 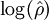 between the two lines (shown in green) serves as an estimate of the potency
between the two lines (shown in green) serves as an estimate of the potency 

