To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Hansen Solubility ParametersHansen Solubility Parameters were developed by Charles Hansen as a way of predicting if one material will dissolve in another and form a solution [1]. They are based on the idea that like dissolves like where one molecule is defined as being 'like' another if it bonds to itself in a similar way. Product highlightSpecifically, each molecule is given three Hansen parameters, each generally measured in
These three parameters can be treated as co-ordinates for a point in three dimensions also known as the Hansen space. The nearer two molecules are in this three dimensional space, the more likely they are to dissolve into each other. To determine if the parameters of two molecules (usually a solvent and a polymer) are within range a value called interaction radius (R0) is given to the substance being dissolved. This value determines the radius of the sphere in Hansen space and it's center is the three Hansen parameters. To calculate the distance (Ra) between Hansen parameters in Hansen space the following formula is used: Combining this with the interaction radius gives the relative energi difference (RED) of the system:
LimitationsThe following limitations were acknowledged by Charles Hansen:
References
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Hansen_Solubility_Parameters". A list of authors is available in Wikipedia. |





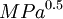 :
:
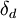 The energy from
The energy from 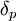 The energy from
The energy from 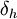 The energy from
The energy from