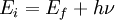To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Inverse photoemission spectroscopyInverse photoemission spectroscopy (IPES) is a surface science technique used to study the unoccupied electronic structure of surfaces, thin films and adsorbates. A well-collimated beam of electrons of a well defined energy (< 20 eV) is directed at the sample. These electrons couple to high-lying unoccupied electronic states and decay to low-lying unocupied states, with a subset of these transitions being radiative. The photons emitted in the decay process are detected and an energy spectrum, photon counts vs. incident electron energy, is generated. Due to the low energy of the incident electrons, their penetration depth is only a few atomic layers, making inverse photoemission a particularly surface sensitive technique. As inverse photoemission probes the electronic states above the Fermi energy of the system, it is a complementary technique to photoemission spectroscopy. Product highlightThe energy of photons (hν) emitted when electrons incident on a substance using an electron beam with a constant energy (Ei) relax to a lower energy unoccupied state (Ef) is given by the conservation of energy as: By measuring Ei and hν, the unoccupied state (Ef) of the surface can be found. Two modes are used for this measurement. One is the isochromat mode, which scans the incident electron energy and keeps the detected photon energy constant. The other is the tunable photon energy mode, or spectrograph mode, which keeps the incident electron energy constant and measures the distribution of the detected photon energy. The latter can also measure the resonant inverse photoemission spectroscopy. Isochromat ModeIn the isochromat mode, the incident electron energy is ramped and the emitted photons are detected at a fixed energy that is determined by the photon detector. Typically, an I2 gas filled Geiger-Müller tube with an entrance window of either SrF2 or CaF2 is used as the photon detector. The combination of window and filling gas determines the detected photon energy, and for I2 gas and either a SrF2 or CaF2 window, the photons energies are ~ 9.5 eV and ~ 9.7 eV, respectively. Spectrograph ModeIn the spectrograph mode, the energy of the incident electron remains fixed and a grating spectrometer is used to the detect the emitted photons over a range of photon energies. A diffraction grating is used to disperse the emitted photons that are in turn detected with a two-dimensional position sensitive detector. Comparison of ModesOne advantage of the spectrograph mode is the ability to acquire IPS spectra over a wide range of photon energies simultaneously. Additionally, the incident electron energy remains fixed which allows better focusing of the electron beam on the sample. Furthermore, by changing the incident electron energy the electronic structure can be studied in great detail. Although the grating spectrometer is very stable over time, the set-up can be very complex and its maintenance can be very expensive. The advantages of isochromat mode are its low cost, simple design and higher count rates. |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Inverse_photoemission_spectroscopy". A list of authors is available in Wikipedia. |