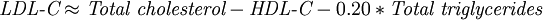To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Low-density lipoproteinLow-density lipoprotein (LDL) belongs to the lipoprotein particle family. Its size is approx. 22 nm and its mass is about 3 million daltons; but, since LDL particles contain a changing number of fatty acids, they actually have a mass and size distribution. Each native LDL particle contains a single apolipoprotein B-100 molecule (Apo B-100, a protein with 4536 amino acid residues) that circles the fatty acids, keeping them soluble in the aqueous environment. In additon, LDL has a highly-hydrophobic core consisting of polyunsaturated fatty acid known as linoleate and about 1500 esterified cholestrol molecules. This core is surrounded by a shell of phospholipids and unesterified cholestrol as well as a single copy of B-100 large protein (514 kD)[1]. Product highlight
FunctionIn general, LDL transports cholesterol and triglycerides from the liver to peripheral tissues. LDL also regulate cholestrol synthesis at these sites. Role in diseaseBecause LDLs transport cholesterol to the arteries and can be retained there by arterial proteoglycans starting the formation of plaques, increased levels are associated with atherosclerosis, and thus heart attack, stroke, and peripheral vascular disease. For this reason, cholesterol inside LDL lipoproteins is often called "bad" cholesterol. This is a misnomer. The cholesterol transported on LDL is the same as cholesterol transported on other lipoprotein particles. The cholesterol itself is not "bad"; rather, it is how and where the cholesterol is being transported, and in what amounts over time, that causes adverse effects. Increasing evidence has revealed that the concentration and size of the LDL particles more powerfully relates to the degree of atherosclerosis progression than the concentration of cholesterol contained within all the LDL particles[citation needed] . The healthiest pattern, though relatively rare, is to have small numbers of large LDL particles and no small particles. Having small LDL particles, though common, is an unhealthy pattern; high concentrations of small LDL particles (even though potentially carrying the same total cholesterol content as a low concentration of large particles) correlates with much faster growth of atheroma, progression of atherosclerosis and earlier and more severe cardiovascular disease events and death. LDL is formed as VLDL lipoproteins lose triglyceride through the action of lipoprotein lipase (LPL) and become smaller and denser, containing a higher proportion of cholesterol. A hereditary form of high LDL is familial hypercholesterolemia (FH). Increased LDL is termed hyperlipoproteinemia type II (after the dated Fredrickson classification). LDL poses a risk for cardiovascular disease when it invades the endothelium and becomes oxidized, since the oxidized form is more easily retained by the proteoglycans. A complex set of biochemical reactions regulates the oxidation of LDL, chiefly stimulated by presence of free-radicals in the endothelium. Nitric oxide down-regulates this oxidation process catalyzed by L-arginine. In a corresponding manner, when there are high levels of asymmetric dimethylarginine in the endothelium, production of nitric oxide is inhibited and more LDL oxidation occurs.Citations required for this paragraph Importance of antioxidantsBecause LDL appears to be harmless until oxidized by free-radicals [2], it is postulated that ingesting antioxidants and minimizing free-radical exposure may reduce LDL's contribution to atherosclerosis, though results are not conclusive. [3] LDL import to the cellWhen a cell requires cholesterol, it synthesises the necessary LDL receptors, and inserts them into the plasma membrane. The LDL receptors diffuse freely until they associate with clathrin-coated pits. LDL particles in the blood stream bind to these extracellular LDL receptors. The clathrin-coated pits then form vesicles that are endocytosed into the cell. After the clathrin coat is shed, the vesicles deliver the LDL and their receptors to early endosomes, onto late endosomes to lysosomes. Here the cholesterol esters in the LDL are hydrolysed. The LDL receptors are recycled back to the plasma membrane. Recommended range; changing targetsThe American Heart Association, NIH, and NCEP provide a set of guidelines for fasting LDL-Cholesterol levels, estimated or measured, and risk for heart disease. As of 2003, these guidelines were:
These guidelines were based on a goal of presumably decreasing death rates from cardiovascular disease to less than 2% to 3% per year or less than 20% to 30% every 10 years. Note that 100 is not considered optimal; less than 100 is optimal, though it is unspecified how much less. Over time, with more clinical research, these recommended levels keep being reduced because LDL reduction, including to abnormally low levels, has been the most effective strategy for reducing cardiovascular death rates in large double blind, randomized clinical trials[citation needed]; far more effective than coronary angioplasty/stenting or bypass surgery. For instance, for people with known atherosclerosis diseases, the 2004 updated American Heart Association, NIH and NCEP recommendations are for LDL levels to be lowered to less than 70 mg/dL, unspecified how much lower. It has been estimated from the results of multiple human pharmacologic LDL lowering trials[citation needed] that LDL should be lowered to about 50 to reduce cardiovascular event rates to near zero. For reference, from longitudinal population studies following progression of atherosclerosis-related behaviors from early childhood into adulthood[citation needed], it has been discovered that the usual LDL in childhood, before the development of fatty streaks, is about 35 mg/dL. However, all the above values refer to chemical measures of lipid/cholesterol concentration within LDL, not LDLipoprotein concentrations, probably not the better approach. Measurement methodsChemical measures of lipid concentration have long been the most-used clinical measurement, not because they have the best correlation with individual outcome, but because these lab methods are less expensive and more widely available. However, there is increasing evidence and recognition of the value of more sophisticated measurements. To be specific, LDL particle number (concentration), and to a lesser extent size, have shown much tighter correlation with atherosclerotic progression and cardiovascular events than is obtained using chemical measures of total LDL concentration contained within the particles. LDL cholesterol concentration can be low, yet LDL particle number high and cardiovascular events rates are high. Also, LDL cholesterol concentration can be relatively high, yet LDL particle number low and cardiovascular events are also low. If LDL particle concentration is tracked against event rates, many other statistical correlates of cardiovascular events, such as diabetes mellitus, obesity, and smoking, lose much of their additive predictive power. LDL subtype patternsLDL particles actually vary in size and density, and studies have shown that a pattern that has more small dense LDL particles—called "Pattern B"—equates to a higher risk factor for coronary heart disease (CHD) than does a pattern with more of the larger and less dense LDL particles ("Pattern A"). This is because the smaller particles are more easily able to penetrate the endothelium. "Pattern I," meaning "intermediate," indicates that most LDL particles are very close in size to the normal gaps in the endothelium (26 nm). The correspondence between Pattern B and CHD has been suggested by some in the medical community to be stronger than the correspondence between the LDL number measured in the standard lipid profile test. Tests to measure these LDL subtype patterns have been more expensive and not widely available, so the common lipid profile test has been used more commonly. The lipid profile does not measure LDL level directly but instead estimates it via the Friedewald equation [4] [5] using levels of other cholesterol such as HDL:
There are limitations to this method, most notably that samples must be obtained after a 12 to 14 h fast and that LDL-C cannot be calculated if plasma triglyceride is >4.52 mmol/L (400 mg/dL). Even at LDC-L levels 2.5 to 4.5 mmol/L, this formula is considered to be inaccurate.[6] If both total cholesterol and triglyceride levels are elevated then a modified formula may be used
This formula provides an approximation with fair accuracy for most people, assuming the blood was drawn after fasting for about 14 hours or longer. (However, the concentration of LDL particles, and to a lesser extent their size, has far tighter correlation with clinical outcome than the content of cholesterol with the LDL particles, even if the LDL-C estimation is about correct.) There has also been noted a correspondence between higher triglyceride levels and higher levels of smaller, denser LDL particles and alternately lower triglyceride levels and higher levels of the larger, less dense LDL. [7] [5] However, cholesterol and lipid assays, as outlined above were never promoted because they worked the best to identify those more likely to have problems, but simply because they used to be far less expensive, by about 50-fold, than measured lipoprotein particle concentrations and subclass analysis. With continued research, decreasing cost, greater availability and wider acceptance of other "lipoprotein subclass analysis" assay methods, including NMR spectroscopy, research studies have continued to show a stronger correlation between human clinically obvious cardiovascular event and quantitatively-measured particle concentrations. Lowering LDLThe mevalonate pathway serves as the basis for the biosynthesis of many molecules, including cholesterol. 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG CoA reductase) is an essential component in the pathway. Pharmaceutical The use of statins (HMG-CoA reductase inhibitors) is effective against high levels of LDL cholesterol. Statins inhibit the enzyme HMG-CoA reductase in the liver, which stimulates LDL receptors, resulting in an increased clearance of LDL. Dietary Insulin induces HMG-CoA reductase activity, whereas glucagon downregulates it.[8] While glucagon production is stimulated by dietary protein ingestion, insulin production is stimulated by dietary carbohydrate. The rise of insulin is, in general, determined by the unfolding of carbohydrates into glucose during the process of digestion. Glucagon levels are very low when insulin levels are high. Lowering the blood lipid concentration of triglycerides, otherwise known as very low-density lipoprotein (VLDL), helps lower the amount of LDL, because VLDL gets converted in the bloodstream into LDL. Fructose, a component of sucrose as well as high-fructose corn syrup, upregulates hepatic VLDL synthesis [9]. Niacin (B3), which blocks breakdown of fats, also lowers VLDL and, as a consequence, LDL. It comes with the added benefit of increasing hi-density lipoprotein, HDL, the so-called 'good' cholesterol.[citation needed] Citations
References
See also
Categories: Lipid disorders | Lipoproteins |
|||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Low-density_lipoprotein". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||