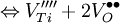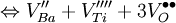To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Schottky defectA Schottky defect is a type of point defect in a crystal lattice named for Walter H. Schottky. The defect forms when oppositely charged ions leave their lattice sites, creating vacancies. These vacancies are formed in stoichiometric units, to maintain an overall neutral charge in the ionic solid. The vacancies are then free to move about as their own entities. Each vacancy is a separate Schottky defect.Normally these defects will lead to a decrease in the density of the crystal.The following are the chemical equations in Kröger-Vink Notation for the formation of Schottky defects in TiO2 and BaTiO3. Product highlightØ
See also |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Schottky_defect". A list of authors is available in Wikipedia. |