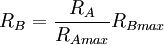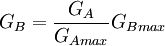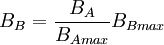To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Standard illuminantA standard illuminant is a profile or spectrum of visible light which is published in order to allow images or colours recorded under different lighting to be compared. Product highlight
CIE illuminantsThe International Commission on Illumination (usually abbreviated CIE for its French name) is the body responsible for publishing all of the well known standard illuminants. Each of these is known by a letter or by a letter-number combination. Illuminant AThe illuminant A has the profile of a black body radiator at 2856 K. It is used to represent incandescent lighting such as a filament lamp. Illuminant series CThe C series of illuminants are based on filtering illuminant A. They are generally considered to be a poor approximation of any common light source and their use is not recommended. Illuminant series DThe D series of illuminants are constructed to represent natural daylight. They are difficult to produce artificially. The number in the name of a D series illuminant represents the correlated colour temperature (CCT) of the source; for example, illuminant D50 has a CCT of 5000 K, and D65 has one of 6500 K. Illuminant series FThe F series of illuminants represent various types of fluorescent lighting. Illuminant F2 represents cool white fluorescent; F11 represents narrow-band fluorescent. F7 and F8 represent daylight fluorescent, and are often used as approximations of D65 and D50, respectively. Standard sourcesIn the past, the CIE published instructions on how to produce the light of a standard illuminant experimentally. These instructions were called standard sources. The CIE no longer publish any standard sources. Experimenters are left to measure to profiles of their sources and compare them to the published spectra of the standards. Although the standard illuminants are now simply tables of arbitrary numbers, many of them are chosen very carefully to be easily reproducible. The D series is a notable exception, as it is based on measurements of natural daylight. White point
The spectrum of a standard illuminant, like any other profile of light, can be converted into tristimulus coordinates. The set of three tristimulus coordinates of an illuminant is called a white point. If the profile is normalised, then the white point can equivalently be expressed as a pair of chromaticity coordinates. If an image is recorded in tristimulus coordinates (or in values which can be converted to and from them), then the white point of the illuminant used gives the maximum value of the tristimulus coordinates that will be recorded at any point in the image, in the absence of fluorescence. It is called the white point of the image. The process of calculating the white point discards a great deal of information about the profile of the illuminant, and so although it is true that for every illuminant the exact white point can be calculated, it is not the case that knowing the white point of an image alone tells you a great deal about the illuminant that was used to record it. White point conversionIf the colour of an object is recorded under one illuminant, then it is possible to estimate the colour of that object under another illuminant, given only the white points of the two illuminants. Expressing colour as tristimulus coordinates R, G and B, then converting from illuminant A to illuminant B:
where RB is the tristimulus value R under illuminant B, and RBmax is the tristimulus white point of illuminant B, etc. This is however, only a very rough estimation of the colour under another illuminant. A slightly better approximation is possible using a Bradford Transform. To truly calculate the colour of an object under another illuminant, it is necessary to record multi-spectral or hyper-spectral colour information. |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Standard_illuminant". A list of authors is available in Wikipedia. |