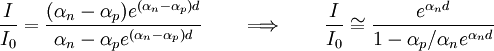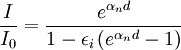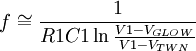To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Townsend discharge
The Townsend discharge is a gas ionisation process where an initially very small amount of free electrons, accelerated by a sufficiently strong electric field, give rise to electrical conduction through a gas by avalanche multiplication: when the number of free charges drops or the electric field weakens, the phenomena ceases. It is a process characterized by very low current densities: in common gas filled tubes, typical magnitude of currents flowing during this process range from about 10 − 18A to about 10 − 5A, while applied voltages are almost constant. Subsequent transition to ionisation processes of dark discharge, glow discharge, and finally to arc discharge are driven by increasing current densities: in all these discharge regimes, the basic mechanism of conduction is avalanche breakdown. Townsend discharge is named after John Sealy Townsend. Product highlight
Quantitative description of the phenomenaThe basic set-up of the experiments investigating ionisation discharges in gases consist of a plane parallel plate capacitor filled with a gas and a continuous current high voltage source connected between its terminals: the terminal at the lower voltage potential is named cathode while the other is named anode. Forcing the cathode to emit electrons (eg. by irradiating it with a X-ray source), Townsend found that the current I flowing into the capacitor depends on the electric field between the plates in such a way that gas ions seems to multilply as they moved between them. He observed currents varying over ten or more orders of magnitude while the applied voltage was virtually constant: the experimental data obtained from his (and his school) first experiments is described by the following formula where
The almost constant voltage between the plates is equal to the breakdown voltage needed to create a self-sustaining avalanche: it decreases when the current reaches the glow discharge regime. Subsequent experiments revealed that the current I rises faster than predicted by the above formula as the distance d increases: two different effects were considered in order to explain the physics of the phenomena and to be able to do a precise quantitative calculation. Gas ionisation caused by motion of positive ionsTownsend put forward the natural hypotesis that also positive ions produce ion pairs, introducing a coefficient αp expressing the number of ion pairs generated per unit length by a positive ion (cation) moving from cathode to anode. The following formula was found since β < < α, in very good agreement with experiments. Cathode emission caused by impact of ionsTownsend and Holst and Oosterhuis also put forward an alternative hypothesis, considering augmented emission of electrons by cathode caused by positive ions impact, introducing Townsends second ionization coefficient εi, the average number of electrons released from a surface by an incident positive ion, and working out the following formula: These two formulas may be thought as describing limiting cases of the effective behavior of the process: note that they can be used to well describe the same experimental results. Other formulas describing, various intermediate behaviors, are found in the literature, particularly in reference 1 and citations therein. Applications
See also
References
Categories: Plasma physics | Electrical breakdown | Ions |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Townsend_discharge". A list of authors is available in Wikipedia. |





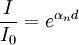
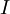 is the current flowing in the device
is the current flowing in the device
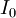 is the
is the 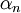 is the first Townsend ionisation coefficient, expressing the number of
is the first Townsend ionisation coefficient, expressing the number of 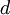 is the distance between the plates of the device
is the distance between the plates of the device