To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Van 't Hoff factorIn physical chemistry, the van 't Hoff factor i is the number of moles of solute actually in solution per mole of solid solute added. Equivalently, i refers to the ratio of true molecular mass to calculated molecular methods by colligative methods. It is named after Jacobus Henricus van 't Hoff, the winner of the first Nobel Prize in chemistry. The van 't Hoff factor is important in quantitative analysis of colligative properties, such as vapor pressure and freezing point depression. Product highlight
Physical significance of iIn NaCl for example, i = 2 indicates that by using the colligative properties equations without the i factor, we're actually calculating the molar mass of each of the Na+ and Cl- ions in solution. The molar mass of NaCl should be twice that number because it originally dissociated to produce one Na+ ion and one Cl- ion per formula unit of NaCl; the sum of the masses of the two ions should yield the mass of one formula unit of NaCl. Note that even if the mass of Na+ and Cl- weren't equal (which is the case), we're only calculating the average mass of the two particles using our colligative properties analysis. Hence multiplying that average number by two, we would be calculating the molar mass of NaCl.
Relation to degree of dissociation
Where α is the degree of association/ dissociation and n is the number of particles produced during association or dissociation per molecule or formula unit of the solute. Illustrative examples
See also
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Van_'t_Hoff_factor". A list of authors is available in Wikipedia. |





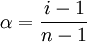
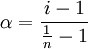
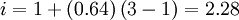 , as each molecule results in one sulfate ion and two hydronium ions. That same result could be achieved by realizing that, for each 100 molecules of acid, 36 are kept and 64 are ionized, resulting in 192 ions and 36 molecules, adding up to a total of 228 particles. Therefore, there would be 2.28 times as many ions as there were acid molecules before.
, as each molecule results in one sulfate ion and two hydronium ions. That same result could be achieved by realizing that, for each 100 molecules of acid, 36 are kept and 64 are ionized, resulting in 192 ions and 36 molecules, adding up to a total of 228 particles. Therefore, there would be 2.28 times as many ions as there were acid molecules before.


