To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Calorimeter constantA calorimeter constant (denoted Ccal) is a constant that quantifies the heat capacity of a calorimeter. It may be calculated by applying a known amount of heat to the calorimeter and measuring the calorimeter's corresponding change in temperature. In SI units, the calorimeter constant is then calculated by dividing the change in heat (ΔH) in joules by the change in temperature (ΔT) in kelvins or degrees Celsius: Product highlightThe calorimeter constant is usually presented in units of joules per degree Celsius (J/°C) or joules per kelvin (J/K). Every calorimeter has a unique calorimeter constant. UsesThe calorimeter constant is used in constant pressure calorimetry to calculate the amount of heat required to raise the contents of the calorimeter by a change in temperature. ExampleFor example, to determine the change in enthalpy in a neutralization reaction (ΔHneutralization), a known amount of basic solution may be placed in a calorimeter, and the temperature of this solution alone recorded. Then, a known amount of acidic solution may be added and the change in temperature measured using a thermometer. The difference in temperature (ΔT, in units K or °C) may be calculated by subtracting the initial temperature from the final temperature. The enthalpy of neutralization ΔHneutralization may then be calculated according to the following equation:
Regardless of the specific chemical process, with a known calorimeter constant and a known change in temperature the heat added to the system may be calculated by multiplying the calorimeter constant by that change in temperature. Categories: Thermochemistry | Thermodynamics |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Calorimeter_constant". A list of authors is available in Wikipedia. |


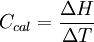



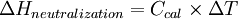 .
.


