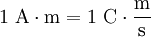To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Ampere-meterThe ampere-metre which has the symbol A m, A-m, or A·m is the SI unit for pole strength in a magnet. Product highlight
DerivationEinstein proved that a magnetic field is the relativistic part of an electric field. This means that while an electric field acts between charges, a magnetic field acts between moving charges (as a charge moves through space more quickly and through time more slowly, its electromagnetic force becomes more magnetic and less electric). Therefore, the pole strength is the product of charge and velocity.
UsefulnessFew calculations actually involve the strength of a pole in ampere-metres because a single magnetic pole has never been isolated. Magnets are dipoles which require more complicated calculations than monopoles. However, the strength of a magnetic field is measured in teslas and one tesla is one newton per ampere-metre which confirms that the unit for pole strength is indeed the ampere-metre. MisconceptionsCharge is usually thought of as a scalar or a quantum number while velocity is a vector. In fact, charge is a vector whose direction is determined by the spin of a particle, but macroscopic calculations are not concerned with this direction and define charge to be along an axis so that the direction of another vector is really its direction relative to charge. A vector pointing along an axis becomes one-dimensional, or identical to a scalar. The idea that magnetic forces act on moving charges is clear in an electromagnet but not obvious in a permanent magnet. In fact, all substances have charges moving in them all of the time, one of the difficulties in reaching absolute zero. In most substances, all of the magnetic fields produced by this motion cancel each other out, but magnetic substances have more proper alignment. At the microscopic level, many molecules are magnets — hydrogen fluoride for example is a dipole. Polarity is most often expressed in terms of electronegativity but the strength of its poles could be expressed in ampere-metres as well. References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Ampere-meter". A list of authors is available in Wikipedia. |