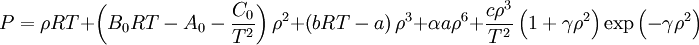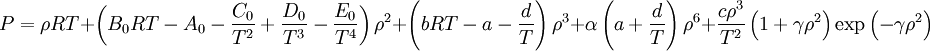To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Benedict-Webb-Rubin equationThe Benedict-Webb-Rubin equation (BWR) is an equation of state used in fluid dynamics. Product highlightThe original BWRThe BWRS Equation of StateA modification of the Benedict-Webb-Rubin equation of state by Professor Kenneth E. Starling of the University of Oklahoma.
Values of the various parameters for 15 substances can be found in: K.E. Starling, Fluid Properties for Light Petroleum Systems. Gulf Publishing Company (1973).
References
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Benedict-Webb-Rubin_equation". A list of authors is available in Wikipedia. |