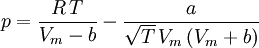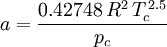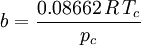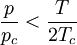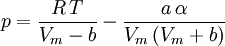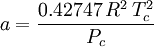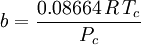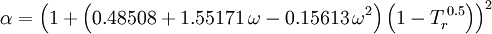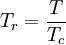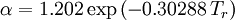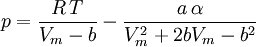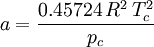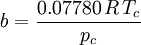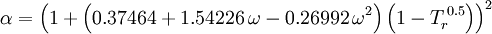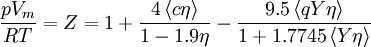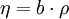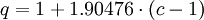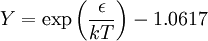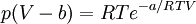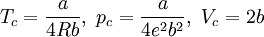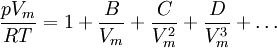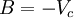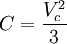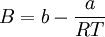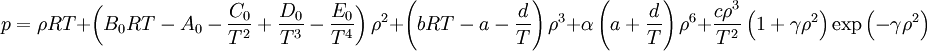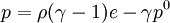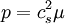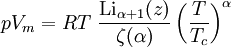To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Equation of state
In physics and thermodynamics, an equation of state is a relation between state variables.[1] More specifically, an equation of state is a thermodynamic equation describing the state of matter under a given set of physical conditions. It is a constitutive equation which provides a mathematical relationship between two or more state functions associated with the matter, such as its temperature, pressure, volume, or internal energy. Equations of state are useful in describing the properties of fluids, mixtures of fluids, solids, and even the interior of stars. Product highlight
OverviewThe most prominent use of an equation of state is to predict the state of gases and liquids. One of the simplest equations of state for this purpose is the ideal gas law, which is roughly accurate for gases at low pressures and high temperatures. However, this equation becomes increasingly inaccurate at higher pressures and lower temperatures, and fails to predict condensation from a gas to a liquid. Therefore, a number of much more accurate equations of state have been developed for gases and liquids. At present, there is no single equation of state that accurately predicts the properties of all substances under all conditions. In addition to predicting the behavior of gases and liquids, there are also equations of state for predicting the volume of solids, including the transition of solids from one crystalline state to another. There are equations that model the interior of stars, including neutron stars. A related concept is the perfect fluid equation of state used in cosmology. HistoricalBoyle's law (1662)Boyle's Law was perhaps the first expression of an equation of state. In 1662 Robert Boyle, an Irishman, performed a series of experiments employing a J-shaped glass tube, which was sealed on one end. Mercury was added to the tube, trapping a fixed quantity of air in the short, sealed end of the tube. Then the volume of gas was carefully measured as additional mercury was added to the tube. The pressure of the gas could be determined by the difference between the mercury level in the short end of the tube and that in the long, open end. Through these experiments, Boyle noted that the gas volume varied inversely with the pressure. In mathematical form, this can be stated as:
The above relationship has also been attributed to Edme Mariotte and is sometimes referred to as Mariotte's law. However, Mariotte's work was not published until 1676. Charles's law or Law of Charles and Gay-Lussac (1787)In 1787 the French physicist Jacques Charles found that oxygen, nitrogen, hydrogen, carbon dioxide, and air expand to the same extent over the same 80 kelvin interval. Later, in 1802, Joseph Louis Gay-Lussac published results of similar experiments, indicating a linear relationship between volume and temperature:
Dalton's law of partial pressures (1801)Dalton's Law of Partial Pressure: The pressure of a mixture of gases is equal to the sum of the pressures of all of the constituent gases alone. Mathematically, this can be represented for n species as:
The ideal gas law (1834)In 1834 Émile Clapeyron combined Boyle's Law and Charles' law into the first statement of the ideal gas law. Initially the law was formulated as pVm=R(TC+267) (with temperature expressed in degrees Celsius). However, later work revealed that the number should actually be closer to 273.2, and then the Celsius scale was defined with 0 °C = 273.15 K, giving:
Van der Waals equation of stateIn 1873, J. D. van der Waals introduced the first equation of state derived by the assumption of a finite volume occupied by the constituent molecules.[2] His new formula revolutionized the study of equations of state, and was most famously continued via the Redlich-Kwong equation of state and the Soave modification of Redlich-Kwong. Major equations of stateIn the following equations the variables are defined as follows. Any consistent set of units may be used, although SI units are preferred. Absolute temperature refers to use of the Kelvin (K) or Rankine (°R) temperature scales, with zero being absolute zero.
Classical ideal gas lawThe classical ideal gas law may be written:
The ideal gas law may also be expressed as follows
where ρ is the density, γ = Cp / Cv is the adiabatic index (ratio of specific heats), e = CvT is the internal energy per unit mass (the "specific internal energy"), Cv is the specific heat at constant volume, and Cp is the specific heat at constant pressure. Cubic equations of stateVan der Waals equation of stateThe Van der Waals equation of state may be written:
Where a and b are constants that depend on the specific material. They can be calculated from the critical properties as: Also written as Proposed in 1873, the van der Waals equation of state was one of the first to perform markedly better than the ideal gas law. In this landmark equation a is called the attraction parameter and b the repulsion parameter or the effective molecular volume. While the equation is definitely superior to the ideal gas law and does predict the formation of a liquid phase, the agreement with experimental data is limited for conditions where the liquid forms. While the van der Waals equation is commonly referenced in text-books and papers for historical reasons, it is now obsolete. Other modern equations of only slightly greater complexity are much more accurate. The van der Waals equation may be considered as the ideal gas law, “improved” due to two independent reasons:
Redlich-Kwong equation of stateIntroduced in 1949 the Redlich-Kwong equation of state was a considerable improvement over other equations of the time. It is still of interest primarily due to its relatively simple form. While superior to the van der Waals equation of state, it performs poorly with respect to the liquid phase and thus cannot be used for accurately calculating vapor-liquid equilibria. However, it can be used in conjunction with separate liquid-phase correlations for this purpose. The Redlich-Kwong equation is adequate for calculation of gas phase properties when the ratio of the pressure to the critical pressure (reduced pressure) is less than about one-half of the ratio of the temperature to the critical temperature (reduced temperature): Soave modification of Redlich-KwongWhere ω is the acentric factor for the species. for hydrogen: In 1972 Soave replaced the a/√(T) term of the Redlich-Kwong equation with a function α(T,ω) involving the temperature and the acentric factor. The α function was devised to fit the vapor pressure data of hydrocarbons and the equation does fairly well for these materials. Note especially that this replacement changes the definition of a slightly, as the Tc is now to the second power. Peng-Robinson equation of statewhere, ω is the acentric factor of the species and R is the universal gas constant. The Peng-Robinson equation was developed in 1976 in order to satisfy the following goals:[3]
For the most part the Peng-Robinson equation exhibits performance similar to the Soave equation, although it is generally superior in predicting the liquid densities of many materials, especially nonpolar ones. The departure functions of the Peng-Robinson equation are given on a separate article. Elliott, Suresh, Donohue equation of stateThe Elliott, Suresh, and Donohue (ESD) equation of state was proposed in 1990. The equation seeks to correct a shortcoming in the Peng-Robinson EOS in that there was an inaccuracy in the van der Waals repulsive term. The EOS accounts for the effect of the shape of a non-polar molecule and can be extended to polymers with the addition of an extra term (not shown). The EOS itself was developed through modeling computer simulations and should capture the essential physics of the size, shape, and hydrogen bonding. where:
Non-cubic equations of stateDieterici equation of stateWhere a is associated with the interaction between molecules and b takes into account the finite size of the molecules, similarly to the Van der Waals equation. The reduced coordinates are: Virial equations of stateVirial equation of stateAlthough usually not the most convenient equation of state, the virial equation is important because it can be derived directly from statistical mechanics. If appropriate assumptions are made about the mathematical form of intermolecular forces, theoretical expressions can be developed for each of the coefficients. In this case B corresponds to interactions between pairs of molecules, C to triplets, and so on. Accuracy can be increased indefinitely by considering higher order terms. It can also be used to work out the Boyle Temperature (the temperature at which B = 0 and ideal gas laws apply) from a and b from the Van der Waals equation of state. If you use the value for B shown below; The BWRS equation of statewhere
Values of the various parameters for 15 substances can be found in: K.E. Starling, Fluid Properties for Light Petroleum Systems. Gulf Publishing Company (1973). Other equations of state of interestStiffened equation of stateWhen considering water under very high pressures (typical applications are underwater nuclear explosions, sonic shock lithotripsy, and sonoluminescence) the stiffened equation of state is often used: where e is the internal energy per unit mass, γ is an empirically determined constant typically taken to be about 6.1, and p0 is another constant, representing the molecular attraction between water molecules. The magnitude of the correction is about 2 gigapascals (20000 atmospheres). The equation is stated in this form because the speed of sound in water is given by c2 = γ(p + p0) / ρ. Thus water behaves as though it is an ideal gas that is already under about 20000 atmospheres (2 GPa) pressure, and explains why water is commonly assumed to be incompressible: when the external pressure changes from 1 atmosphere to 2 atmospheres (100 kPa to 200 kPa), the water behaves as an ideal gas would do when changing from 20001 to 20002 atmospheres (2000.1 MPa to 2000.2 MPa). This equation mispredicts the specific heat capacity of water but few alternatives are available for severely nonisentropic processes such as strong shocks. Ultrarelativistic equation of stateAn ultrarelativistic fluid has equation of state where p is the pressure, μ is the energy density, and cs is the speed of sound. Ideal Bose equation of stateThe equation of state for an ideal Bose gas is where α is an exponent specific to the system (e.g. in the absence of a potential field, α=3/2), z is exp(μ/kT) where μ is the chemical potential, Li is the polylogarithm, ζ is the Riemann zeta function, and Tc is the critical temperature at which a Bose-Einstein condensate begins to form. Equations of state for solidsSee alsoBibliography
References
Categories: Thermodynamics | Chemical engineering | Fluid mechanics | Phases of matter |
|||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Equation_of_state". A list of authors is available in Wikipedia. |





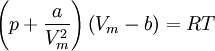 , note that
, note that 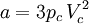
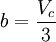
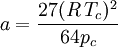
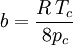
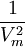 .
.