To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Internal energy
The internal energy is a thermodynamic potential and for a closed thermodynamic system held at constant entropy, it will be minimized. One can also calculate the internal energy of electromagnetic or blackbody radiation. It is a state function of a system, an extensive quantity. The SI unit of energy is the joule although other historical, conventional units are still in use, such as the (small and large) calorie for heat. Product highlight
OverviewInternal energy does not include the translational or rotational kinetic energy of a body as a whole. It also does not include the relativistic mass-energy equivalent E = mc2. It excludes any potential energy a body may have because of its location in external gravitational or electrostatic field, although the potential energy it has in a field due to an induced electric or magnetic dipole moment does count, as does the energy of deformation of solids (stress-strain). The principle of equipartition of energy in classical statistical mechanics states that each molecular degree of freedom receives 1/2 kT of energy, a result which was modified when quantum mechanics explained certain anomalies; e.g., in the observed specific heats of crystals (when hν > kT). For monatomic helium and other noble gases, the internal energy consists only of the translational kinetic energy of the individual atoms. Monatomic particles, of course, do not (sensibly) rotate or vibrate, and are not electronically excited to higher energies except at very high temperatures. From the standpoint of statistical mechanics, the internal energy is equal to the ensemble average of the total energy of the system. CompositionInternal energy – the sum of all microscopic forms of energy of a system. It is related to the molecular structure and the degree of molecular activity and may be viewed as the sum of kinetic and potential energies of the molecules; it is comprised of the following types of energies:[1]
The first law of thermodynamicsThe internal energy is essentially defined by the first law of thermodynamics which states that energy is conserved: where
The first law may be equivalently in infinitesimal terms as: where the terms now represent infinitesimal amounts of the respective quantities. The d before the internal energy function indicates that it is an exact differential. In other words it is a state function or a value which can be assigned to the system. On the other hand, the δ's before the other terms indicate that they describe increments of energy which are not state functions but rather they are processes by which the internal energy is changed. (See the discussion in the first law article.) From a microscopic point of view, the internal energy may be found in many different forms. For a gas it may consist almost entirely of the kinetic energy of the gas molecules. It may also consist of the potential energy of these molecules in a gravitational, electric, or magnetic field. For any material, solid, liquid or gaseous, it may also consist of the potential energy of attraction or repulsion between the individual molecules of the material. Expressions for the internal energyStrictly speaking, the internal energy cannot be precisely measured. This is because only changes in the internal energy can be measured, and the total internal energy of a given system is the difference between the internal energy of the system and the internal energy of the same system at absolute zero temperature. Since absolute zero cannot be attained, the total internal energy cannot be precisely measured. The same is true of other thermodynamic parameters such as entropy and the chemical potential. The internal energy may be expressed in terms of other thermodynamic parameters. Each term is composed of an intensive variable (a generalized force) and its conjugate infinitesimal extensive variable (a generalized displacement). For example, for a non-viscous fluid, the mechanical work done on the system may be related to the pressure p and volume V. The pressure is the intensive generalized force, while the volume is the extensive generalized displacement: Taking the default direction of work, W, to be from the working fluid to the surroundings,
Taking the default direction of heat transfer, Q, to be into the working fluid and assuming a reversible process, we have
Although the internal energy is not exactly measurable, it may be expressed in terms of other similarly unmeasurable quantities. Using the above two equations in the first law of thermodynamics to construct one possible expression for the internal energy of a closed system gives: The internal energy function may be written as U(S,V) in which case it follows that, since U, S, and V are extensive From Euler's homogeneous function theorem we may now write the internal energy as: If the (non-viscous) fluid gains energy from the addition of particles, we add the chemical energy term:
For an elastic substance the mechanical term must be replaced by the more general expression involving the stress σij and strain where Einstein notation has been used for the tensors, in which there is a summation over all repeated indices in the product term. For a linearly elastic material, the stress can be related to the strain by: and the Euler theorem yields for the internal energy (Landau & Lifshitz 1986): References
See alsoCategories: Thermodynamics | Statistical mechanics |
|||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Internal_energy". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||





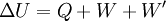
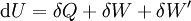
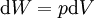 .
.
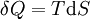 .
.
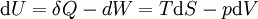
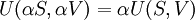
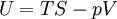
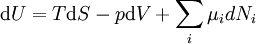
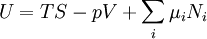 .
.
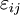 . The infinitesimal statement is:
. The infinitesimal statement is: