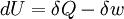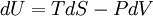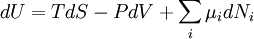To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
First law of thermodynamics
In thermodynamics, the first law of thermodynamics is an expression of the more universal physical law of the conservation of energy. Product highlight
StatementsSuccinctly, the first law of thermodynamics states:
DescriptionThe first law of thermodynamics basically states that a thermodynamic system can store or hold energy and that this internal energy is conserved. Heat is a process by which energy is added to a system from a high-temperature source, or lost to a low-temperature sink. In addition, energy may be lost by the system when it does mechanical work on its surroundings, or conversely, it may gain energy as a result of work done on it by its surroundings. The first law states that this energy is conserved: The change in the internal energy is equal to the amount added by heating minus the amount lost by doing work on the environment. The first law can be stated mathematically as: where dU is a small increase in the internal energy of the system, δQ is a small amount of heat added to the system, and δW is a small amount of work done by the system. As an analogy, if heat were money, then we could say that any change in our savings (dU) is equal to the money we put in (δQ) minus the money we spend (δw). The δ's before the heat and work terms are used to indicate that they describe an increment of energy which is to be interpreted somewhat differently than the dU increment of internal energy. Work and heat are processes which add or subtract energy, while the internal energy U is a particular form of energy associated with the system. Thus the term "heat energy" for δQ means "that amount of energy added as the result of heating" rather than referring to a particular form of energy. Likewise, the term "work energy" for δw means "that amount of energy lost as the result of work". The most significant result of this distinction is the fact that one can clearly state the amount of internal energy possessed by a thermodynamic system, but one cannot tell how much energy has flowed into or out of the system as a result of its being heated or cooled, nor as the result of work being performed on or by the system. The first explicit statement of the first law of thermodynamics was given by Rudolf Clausius in 1850: "There is a state function E, called ‘energy’, whose differential equals the work exchanged with the surroundings during an adiabatic process." Note that the above formulation is favored by engineers and physicists. Chemists prefer a second form, in which the work term δw is defined as the work done on the system, and therefore insert a plus sign in the above equation before the work term. This article will use the first definition exclusively. Mathematical formulationThe mathematical statement of the first law is given by: where dU is the infinitesimal increase in the internal energy of the system, δQ is the infinitesimal amount of heat added to the system, and δw is the infinitesimal amount of work done by the system. The infinitesimal heat and work are denoted by δ rather than d because, in mathematical terms, they are not exact differentials. In other words, they do not describe the state of any system. The integral of an inexact differential depends upon the particular "path" taken through the space of thermodynamic parameters while the integral of an exact differential depends only upon the initial and final states. If the initial and final states are the same, then the integral of an inexact differential may or may not be zero, but the integral of an exact differential will always be zero. The path taken by a thermodynamic system through state space is known as a thermodynamic process. An expression of the first law can be written in terms of exact differentials by realizing that the work that a system does is equal to its pressure times the infinitesimal change in its volume. In other words δw = PdV where P is pressure and V is volume. For a reversible process, the total amount of heat added to a system can be expressed as δQ = TdS where T is temperature and S is entropy. For a reversible process, the first law may now be restated: In the case where the number of particles in the system is not necessarily constant and may be of different types, the first law is written: where dNi is the (small) number of type-i particles added to the system, and μi is the amount of energy added to the system when one type-i particle is added, where the energy of that particle is such that the volume and entropy of the system remains unchanged. μi is known as the chemical potential of the type-i particles in the system. The statement of the first law for reversible processes, using exact differentials is now: A useful idea from mechanics is that the energy gained by a particle is equal to the force applied to the particle multiplied by the displacement of the particle while that force is applied. Now consider the first law without the heating term: dU = PdV. The pressure P can be viewed as a force (and in fact has units of force per unit area) while dV is the displacement (with units of distance times area). We may say, with respect to this work term, that a pressure difference forces a transfer of volume, and that the product of the two (work) is the amount of energy transferred as a result of the process. It is useful to view the TdS term in the same light: With respect to this heat term, a temperature difference forces a transfer of entropy, and the product of the two (heat) is the amount of energy transferred as a result of the process. Here, the temperature is known as a "generalized" force (rather than an actual mechanical force) and the entropy is a generalized displacement. Similarly, a difference in chemical potential between groups of particles in the system forces a trasfer of particles, and the corresponding product is the amount of energy transferred as a result of the process. For example, consider a system consisting of two phases: liquid water and water vapor. There is a generalized "force" of evaporation which drives water molecules out of the liquid. There is a generalized "force" of condensation which drives vapor molecules out of the vapor. Only when these two "forces" (or chemical potentials) are equal will there be equilibrium, and the net transfer will be zero. The two thermodynamic parameters which form a generalized force-displacement pair are termed "conjugate variables". The two most familiar pairs are, of course, pressure-volume, and temperature-entropy. Types of thermodynamic processesPaths through the space of thermodynamic variables are often specified by holding certain thermodynamic variables constant. It is useful to group these processes into pairs, in which each variable held constant is one member of a conjugate pair. The pressure-volume conjugate pair is concerned with the transfer of mechanical or dynamic energy as the result of work.
The temperature-entropy conjugate pair is concerned with the transfer of thermal energy as the result of heating.
The above have all implicitly assumed that the boundaries are also impermeable to particles. We may assume boundaries that are both rigid and thermally insulating, but are permeable to one or more types of particle. Similar considerations then hold for the (chemical potential)-(particle number) conjugate pairs.
See also
Categories: Laws of thermodynamics | Atmospheric thermodynamics |
||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "First_law_of_thermodynamics". A list of authors is available in Wikipedia. |