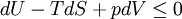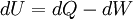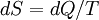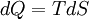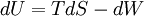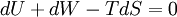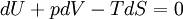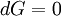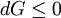To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Combined law of thermodynamics
In thermodynamics, the combined law of thermodynamics is a mathematical summation of the first law of thermodynamics and the second law of thermodynamics subsumed into a single concise mathematical statement as shown below: Product highlightHere, U is internal energy, T is temperature, S is entropy, p is pressure, and V is volume. In theoretical structure in addition to the obvious inclusion of the first two laws, the combined law incorporates the implications of the zeroth law, via temperature T, and the third law, through its use of free energy as related to the calculation of chemical affinities near absolute zero. DerivationStarting from the first law, and neglecting differential details: From the second law we have: Hence: By substituting this into the first law, we have: Rearranging we have: Letting dW be pressure-volume work, we have: By assigning the quantity to the left of the equals sign the symbol G, as Willard Gibbs did in 1876, this reduces to the following at thermodynamic equilibrium: Or for a spontaneous process: Thus, this expression is referred to by many as the combined law of thermodynamics; Gibbs showed that deviations of this quantity could be used to predict the direction of various natural chemical processes. |
|||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Combined_law_of_thermodynamics". A list of authors is available in Wikipedia. |