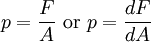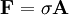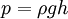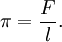To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Pressure
Gauge pressure is the pressure relative to the local atmospheric or ambient pressure. Product highlight
DefinitionFormulaic
Mathematically: where:
Pressure is a scalar quantity, and has SI units of pascals; 1 Pa = 1 N/m2. Pressure is transmitted to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. It is a fundamental parameter in thermodynamics and it is conjugate to volume. Units
The SI unit for pressure is the pascal (Pa), equal to one newton per square metre (N·m-2 or kg·m-1·s-2). This special name for the unit was added in 1971; before that, pressure in SI was expressed simply as N/m2. Non-SI measures such as pound per square inch (psi) and bar are used in some parts of the world. The cgs unit of pressure is the barye (ba), equal to 1 dyn·cm-2. Pressure is sometimes expressed in grams-force/cm2, or as [[kg/cm2]] and the like without properly identifying the force units. But using the names kilogram, gram, kilogram-force, or gram-force (or their symbols) as units of force is expressly forbidden in SI. The technical atmosphere (symbol: at) is 1 kgf/cm2. Some meteorologists prefer the hectopascal (hPa) for atmospheric air pressure, which is equivalent to the older unit millibar (mbar). Similar pressures are given in kilopascals (kPa) in most other fields, where the hecto prefix is rarely used. The unit inch of mercury (inHg, see below) is still used in the United States. Oceanographers usually measure underwater pressure in decibars (dbar) because an increase in pressure of 1 dbar is approximately equal to an increase in depth of 1 meter. Scuba divers often use a manometric rule of thumb: the pressure exerted by ten metres depth of water is approximately equal to one atmosphere. The standard atmosphere (atm) is an established constant. It is approximately equal to typical air pressure at earth mean sea level and is defined as follows:
Because pressure is commonly measured by its ability to displace a column of liquid in a manometer, pressures are often expressed as a depth of a particular fluid (e.g., inches of water). The most common choices are mercury (Hg) and water; water is nontoxic and readily available, while mercury's high density allows for a shorter column (and so a smaller manometer) to measure a given pressure. The pressure exerted by a column of liquid of height h and density ρ is given by the hydrostatic pressure equation p = ρgh. Fluid density and local gravity can vary from one reading to another depending on local factors, so the height of a fluid column does not define pressure precisely. When millimeters of mercury or inches of mercury are quoted today, these units are not based on a physical column of mercury; rather, they have been given precise definitions that can be expressed in terms of SI units. The water-based units still depend on the density of water, a measured, rather than defined, quantity. These manometric units are still encountered in many fields. Blood pressure is measured in millimeters of mercury in most of the world, and lung pressures in centimeters of water are still common. Presently or formerly popular pressure units include the following:
Example reading: 1 Pa = 1 N/m2 = 10−5 bar = 10.197×10−6 at = 9.8692×10−6 atm, etc.
ExamplesAs an example of varying pressures, a finger can be pressed against a wall without making any lasting impression; however, the same finger pushing a thumbtack can easily damage the wall. Although the force applied to the surface is the same, the thumbtack applies more pressure because the point concentrates that force into a smaller area. Pressure is transmitted to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. Unlike stress, pressure is defined as a scalar quantity. The gradient of pressure is called the force density. For gases, pressure is sometimes measured not as an absolute pressure, but relative to atmospheric pressure; such measurements are called gauge pressure (also sometimes spelled gage pressure).[1] An example of this is the air pressure in an automobile tire, which might be said to be "220 kPa", but is actually 220 kPa above atmospheric pressure. Since atmospheric pressure at sea level is about 100 kPa, the absolute pressure in the tire is therefore about 320 kPa. In technical work, this is written "a gauge pressure of 220 kPa". Where space is limited, such as on pressure gauges, name plates, graph labels, and table headings, the use of a modifier in parentheses, such as "kPa (gauge)" or "kPa (absolute)", is permitted. In non-SI technical work, a gauge pressure is sometimes written as "32 psig", though the other methods explained above that avoid attaching characters to the unit of pressure are preferred.[2] Gauge pressure is the relevant measure of pressure wherever one is interested in the stress on storage vessels and the plumbing components of fluidics systems. However, whenever equation-of-state properties, such as densities or changes in densities, must be calculated, pressures must be expressed in terms of their absolute values. For instance, if the atmospheric pressure is 100 kPa, a gas (such as helium) at 200 kPa (gauge) (300 kPa [absolute]) is 50 % more dense than the same gas at 100 kPa (gauge) (200 kPa [absolute]). Focusing on gauge values, one might erroneously conclude the first sample had twice the density of the second. Scalar natureIn a static gas, the gas as a whole does not appear to move. The individual molecules of the gas, however, are in constant random motion. Because we are dealing with an extremely large number of molecules and because the motion of the individual molecules is random in every direction, we do not detect any motion. If we enclose the gas within a container, we detect a pressure in the gas from the molecules colliding with the walls of our container. We can put the walls of our container anywhere inside the gas, and the force per unit area (the pressure) is the same. We can shrink the size of our "container" down to an infinitely small point, and the pressure has a single value at that point. Therefore, pressure is a scalar quantity, not a vector quantity. It has a magnitude but no direction associated with it. Pressure acts in all directions at a point inside a gas. At the surface of a gas, the pressure force acts perpendicular to the surface. A closely related quantity is the stress tensor σ, which relates the vector force F to the vector area A via This tensor may be divided up into a scalar part (pressure) and a traceless tensor part shear. The shear tensor gives the force in directions parallel to the surface, usually due to viscous or frictional forces. The stress tensor is sometimes called the pressure tensor, but in the following, the term "pressure" will refer only to the scalar pressure. TypesExplosion or deflagration pressuresExplosion or deflagration pressures are the result of the ignition of explosible gases, mists, dust/air suspensions, in unconfined and confined spaces. Negative pressuresWhile pressures are generally positive, there are several situations in which a negative pressure may be encountered:
Hydrostatic pressure (head pressure)Hydrostatic pressure is the pressure due to the weight of a fluid. where:
See also Pascal's law. Stagnation pressureStagnation pressure is the pressure a fluid exerts when it is forced to stop moving. Consequently, although a fluid moving at higher speed will have a lower static pressure, it may have a higher stagnation pressure when forced to a standstill. Static pressure and stagnation pressure are related by the Mach number of the fluid. In addition, there can be differences in pressure due to differences in the elevation (height) of the fluid. See Bernoulli's equation (note: Bernoulli's equation only applies for incompressible flow). The pressure of a moving fluid can be measured using a Pitot tube, or one of its variations such as a Kiel probe or Cobra probe, connected to a manometer. Depending on where the inlet holes are located on the probe, it can measure static pressure or stagnation pressure. Surface pressureThere is a two-dimensional analog of pressure -- the lateral force per unit length applied on a line perpendicular to the force. Surface pressure is denoted by π and shares many similar properties with three-dimensional pressure. Properties of surface chemicals can be investigated by measuring pressure/area isotherms, as the two-dimensional analog of Boyle's law, πA = k, at constant temperature. See also
Notes
Categories: Atmospheric thermodynamics | Thermodynamics |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Pressure". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||