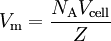To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Molar volumeThe molar volume, symbol Vm,[1] is the volume occupied by one mole of a substance (chemical element or chemical compound) at a given temperature and pressure. It is equal to the molar mass (M) divided by the mass density (ρ). It has the SI unit cubic metres per mole (m³/mol),[1] although it is more practical to use the units cubic decimetres per mole (dm³/mol) for gases and cubic centimetres per mole (cm³/mol) for liquids and solids. The molar volume of a substance can be found by measuring its mass density then applying the relation
For ideal gases, the molar volume is given by the ideal gas equation: this is a good approximation for many common gases at standard temperature and pressure. For crystalline solids, the molar volume can be measured by X-ray crystallography. Product highlight
Ideal gasesThe ideal gas equation can be rearranged to give an expression for the molar volume of an ideal gas:
Hence, for a given temperature and pressure, the molar volume is the same for all ideal gases and is known to the same precision as the gas constant: R = 8.314 472(15) J mol–1 K–1, that is a relative standard uncertainty of 1.7×10–6, according to the 2006 CODATA recommended value.[2] The molar volume of an ideal gas at 100 kPa (1 bar) is
Crystalline solidsThe unit cell volume (Vcell) may be calculated from the unit cell parameters, whose determination is the first step in an X-ray crystallography experiment (the calculation is performed automatically by the structure determination software). This is related to the molar volume by where NA is the Avogadro constant and Z is the number of formula units in the unit cell. The result is normally reported as the "crystallographic density". Molar volume of siliconHigh quality single crystals of ultrapure silicon are routinely made for the electronics industry, and the measurement of the molar volume of silicon, both by X-ray crystallography and by the ratio of molar mass to mass density, has attracted much attention since the pioneering work at NIST by Deslattes et al. (1974).[3] The interest stems from the fact that accurate measurements of the unit cell volume, atomic weight and mass density of a pure crystalline solid provide a direct determination of the Avogadro constant.[4] At present (2006 CODATA recommended value),[5] the precision of the value of the Avogadro constant is limited by the uncertainty in the value of the Planck constant (relative standard uncertainty of 5×10–8).[4][6] The 2006 CODATA recommended value for the molar volume of silicon is 12.058 8349(11)×10–6 m³/mol, with a relative standard uncertainty of 9.1×10–8.[7] References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Molar_volume". A list of authors is available in Wikipedia. |


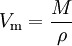 .
.



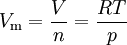 .
.