To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Plasma (physics)
In physics and chemistry, a plasma is typically an ionized gas. Plasma is considered to be a distinct state of matter, apart from gases, because of its unique properties. Ionized refers to presence of one or more free electrons, which are not bound to an atom or molecule. The free electric charges make the plasma electrically conductive so that it responds strongly to electromagnetic fields. Plasma typically takes the form of neutral gas-like clouds (e.g. stars) or charged ion beams, but may also include dust and grains (called dusty plasmas).[1] They are typically formed by heating and ionizing a gas, stripping electrons away from atoms, thereby enabling the positive and negative charges to move more freely. Product highlight
HistoryThis state of matter was first identified in a Crookes tube, and so described by Sir William Crookes in 1879 (he called it "radiant matter").[2] The nature of the Crookes tube "cathode ray" matter was subsequently identified by British physicist Sir J.J. Thomson in 1897,[3] and dubbed "plasma" by Irving Langmuir in 1928,[4] perhaps because it reminded him of a blood plasma.[5] Langmuir wrote:
Common plasmasPlasmas are by far the most common phase of matter in the universe, both by mass and by volume.[6] All the stars are made of plasma, and even the space between the stars is filled with a plasma, albeit a very sparse one (See astrophysical plasma, interstellar medium and intergalactic space). In the solar system, the planet Jupiter accounts for most of the non-plasma, only about 0.1% of the mass and 10−15% of the volume within the orbit of Pluto. Notable plasma physicist Hannes Alfvén also noted that due to their electric charge, very small grains also behave as ions and form part of plasma (see dusty plasmas).
Plasma properties and parameters
Definition of a plasmaAlthough a plasma is loosely described as an electrically neutral medium of positive and negative particles, a more rigorous definition requires three criteria to be satisfied:
Ranges of plasma parametersPlasma parameters can take on values varying by many orders of magnitude, but the properties of plasmas with apparently disparate parameters may be very similar (see plasma scaling). The following chart considers only conventional atomic plasmas and not exotic phenomena like quark gluon plasmas:
Degree of ionizationFor plasma to exist, ionization is necessary. The word "plasma density" by itself usually refers to the electron density, that is, the number of free electrons per unit volume. The degree of ionization of a plasma is the proportion of atoms which have lost (or gained) electrons, and is controlled mostly by the temperature. Even a partially ionized gas in which as little as 1% of the particles are ionized can have the characteristics of a plasma (i.e. respond to magnetic fields and be highly electrically conductive). The degree of ionization, α is defined as α = ni/(ni + na) where ni is the number density of ions and na is the number density of neutral atoms. The electron density is related to this by the average charge state TemperaturesPlasma temperature is commonly measured in kelvins or electronvolts, and is (roughly speaking) a measure of the thermal kinetic energy per particle. In most cases the electrons are close enough to thermal equilibrium that their temperature is relatively well-defined, even when there is a significant deviation from a Maxwellian energy distribution function, for example due to UV radiation, energetic particles, or strong electric fields. Because of the large difference in mass, the electrons come to thermodynamic equilibrium among themselves much faster than they come into equilibrium with the ions or neutral atoms. For this reason the ion temperature may be very different from (usually lower than) the electron temperature. This is especially common in weakly ionized technological plasmas, where the ions are often near the ambient temperature. Based on the relative temperatures of the electrons, ions and neutrals, plasmas are classified as thermal or non-thermal. Thermal plasmas have electrons and the heavy particles at the same temperature i.e. they are in thermal equilibrium with each other. Non thermal plasmas on the other hand have the ions and neutrals at a much lower temperature (normally room temperature) whereas electrons are much "hotter". Temperature controls the degree of plasma ionization. In particular, plasma ionization is determined by the electron temperature relative to the ionization energy (and more weakly by the density) in accordance with the Saha equation. A plasma is sometimes referred to as being hot if it is nearly fully ionized, or cold if only a small fraction (for example 1%) of the gas molecules are ionized (but other definitions of the terms hot plasma and cold plasma are common). Even in a "cold" plasma the electron temperature is still typically several thousand degrees Celsius. Plasmas utilized in plasma technology ("technological plasmas") are usually cold in this sense. Potentials
Since plasmas are very good conductors, electric potentials play an important role.
The potential as it exists on average in the space between charged particles, independent of the question of how it can be measured, is called the plasma potential or the space potential. If an electrode is inserted into a plasma, its potential will generally lie considerably below the plasma potential due to the development of a Debye sheath. Due to the good electrical conductivity, the electric fields in plasmas tend to be very small. This results in the important concept of quasineutrality, which says that it is a very good approximation to assume that the density of negative charges is equal to the density of positive charges over large volumes of the plasma ( The magnitude of the potentials and electric fields must be determined by means other than simply finding the net charge density. A common example is to assume that the electrons satisfy the Boltzmann relation:
Differentiating this relation provides a means to calculate the electric field from the density:
It is, of course, possible to produce a plasma that is not quasineutral. An electron beam, for example, has only negative charges. The density of a non-neutral plasma must generally be very low, or it must be very small, otherwise it will be dissipated by the repulsive electrostatic force. In astrophysical plasmas, Debye screening prevents electric fields from directly affecting the plasma over large distances (ie. greater than the Debye length). But the existence of charged particles causes the plasma to generate and be affected by magnetic fields. This can and does cause extremely complex behavior, such as the generation of plasma double layers, an object that separates charge over a few tens of Debye lengths. The dynamics of plasmas interacting with external and self-generated magnetic fields are studied in the academic discipline of magnetohydrodynamics. MagnetizationA plasma in which the magnetic field is strong enough to influence the motion of the charged particles is said to be magnetized. A common quantitative criterion is that a particle on average completes at least one gyration around the magnetic field before making a collision (ie. ωce / νcoll > 1 where ωce is the "electron gyrofrequency" and νcoll is the "electron collision rate"). It is often the case that the electrons are magnetized while the ions are not. Magnetized plasmas are anisotropic, meaning that their properties in the direction parallel to the magnetic field are different from those perpendicular to it. While electric fields in plasmas are usually small due to the high conductivity, the electric field associated with a plasma moving in a magnetic field is given by E = -v x B (where E is the electric field, v is the velocity, and B is the magnetic field), and is not affected by Debye shielding.[11] Comparison of plasma and gas phasesPlasma is often called the fourth state of matter. It is distinct from other lower-energy states of matter; most commonly solid, liquid, and gas, although it is closely related to the gas phase in that it also has no definite form or volume. Physicists consider a plasma to be more than a gas because of a number of distinct properties including the following:
Complex plasma phenomena
Although the underlying equations governing plasmas are relatively simple, plasma behaviour is extraordinarily varied and subtle: the emergence of unexpected behaviour from a simple model is a typical feature of a complex system. Such systems lie in some sense on the boundary between ordered and disordered behaviour, and cannot typically be described either by simple, smooth, mathematical functions, or by pure randomness. The spontaneous formation of interesting spatial features on a wide range of length scales is one manifestation of plasma complexity. The features are interesting, for example, because they are very sharp, spatially intermittent (the distance between features is much larger than the features themselves), or have a fractal form. Many of these features were first studied in the laboratory, and have subsequently been recognised throughout the universe. Examples of complexity and complex structures in plasmas include: FilamentationThe striations or "stringy" things,[13] seen in many plasmas, like the plasma ball (image above), the aurora,[14] lightning,[15] electric arcs, solar flares,[16] and supernova remnants[17] They are sometimes associated with larger current densities, and are also called magnetic ropes.[18] (See also Plasma pinch) Shocks or double layersNarrow sheets with sharp gradients, such as shocks or double layers which support rapid changes in plasma properties. Double layers involve localised charge separation, which causes a large potential difference across the layer, but does not generate an electric field outside the layer. Double layers separate adjacent plasma regions with different physical characteristics, and are often found in current carrying plasmas. They accelerate both ions and electrons. Electric fields and circuitsQuasineutrality of a plasma requires that plasma currents close on themselves in electric circuits. Such circuits follow Kirchhoff's circuit laws, and possess a resistance and inductance. These circuits must generally be treated as a strongly coupled system, with the behaviour in each plasma region dependent on the entire circuit. It is this strong coupling between system elements, together with nonlinearity, which may lead to complex behaviour. Electrical circuits in plasmas store inductive (magnetic) energy, and should the circuit be disrupted, for example, by a plasma instability, the inductive energy will be released as plasma heating and acceleration. This is a common explanation for the heating which takes place in the solar corona. Electric currents, and in particular, magnetic-field-aligned electric currents (which are sometimes generically referred to as Birkeland currents), are also observed in the Earth's aurora, and in plasma filaments. Cellular structureNarrow sheets with sharp gradients may separate regions with different properties such as magnetization, density, and temperature, resulting in cell-like regions. Examples include the magnetosphere, heliosphere, and heliospheric current sheet. Hannes Alfvén wrote: "From the cosmological point of view, the most important new space research discovery is probably the cellular structure of space. As has been seen, in every region of space which is accessible to in situ measurements, there are a number of 'cell walls', sheets of electric currents, which divide space into compartments with different magnetization, temperature, density, etc ."[19] Critical ionization velocityThe Critical ionization velocity is the relative velocity between an (magnetized) ionized plasma and a neutral gas above which a runaway ionization process takes place. The critical ionization process is a quite general mechanism for the conversion of the kinetic energy of a rapidly streaming gas into ionization and plasma thermal energy. Critical phenomena in general are typical of complex systems, and may lead to sharp spatial or temporal features. Ultracold plasmaIt is possible to create ultracold plasmas, by using lasers to trap and cool neutral atoms to temperatures of 1 mK or lower. Another laser then ionizes the atoms by giving each of the outermost electrons just enough energy to escape the electrical attraction of its parent ion. The key point about ultracold plasmas is that by manipulating the atoms with lasers, the kinetic energy of the liberated electrons can be controlled. Using standard pulsed lasers, the electron energy can be made to correspond to a temperature of as low as 0.1 K, a limit set by the frequency bandwidth of the laser pulse. The ions, however, retain the millikelvin temperatures of the neutral atoms. This type of non-equilibrium ultracold plasma evolves rapidly, and many fundamental questions about its behaviour remain unanswered. Experiments conducted so far have revealed surprising dynamics and recombination behavior that are pushing the limits of our knowledge of plasma physics.[citation needed] One of the metastable states of strongly nonideal plasma is Rydberg matter which forms upon condensation of excited atoms. Non-neutral plasmaThe strength and range of the electric force and the good conductivity of plasmas usually ensure that the density of positive and negative charges in any sizeable region are equal ("quasineutrality"). A plasma that has a significant excess of charge density or that is, in the extreme case, composed of only a single species, is called a non-neutral plasma. In such a plasma, electric fields play a dominant role. Examples are charged particle beams, an electron cloud in a Penning trap, and positron plasmas.[20] Dusty plasma and grain plasmaA dusty plasma is one containing tiny charged particles of dust (typically found in space) that also behaves like a plasma. A plasma containing larger particles is called a grain plasma. Mathematical descriptions
To completely describe the state of a plasma, we would need to write down all the particle locations and velocities, and describe the electromagnetic field in the plasma region. However, it is generally not practical or necessary to keep track of all the particles in a plasma. Therefore, plasma physicists commonly use less detailed descriptions known as models, of which there are two main types: Fluid modelFluid models describe plasmas in terms of smoothed quantities like density and averaged velocity around each position (see Plasma parameters). One simple fluid model, magnetohydrodynamics, treats the plasma as a single fluid governed by a combination of Maxwell's Equations and the Navier Stokes Equations. A more general description is the two-fluid picture, where the ions and electrons are described separately. Fluid models are often accurate when collisionality is sufficiently high to keep the plasma velocity distribution close to a Maxwell-Boltzmann distribution. Because fluid models usually describe the plasma in terms of a single flow at a certain temperature at each spatial location, they can neither capture velocity space structures like beams or double layers nor resolve wave-particle effects. Kinetic modelKinetic models describe the particle velocity distribution function at each point in the plasma, and therefore do not need to assume a Maxwell-Boltzmann distribution. A kinetic description is often necessary for collisionless plasmas. There are two common approaches to kinetic description of a plasma. One is based on representing the smoothed distribution function on a grid in velocity and position. The other, known as the particle-in-cell (PIC) technique, includes kinetic information by following the trajectories of a large number of individual particles. Kinetic models are generally more computationally intensive than fluid models. The Vlasov equation may be used to describe how a system of particles evolves in an electromagnetic environment. Common artificial plasmaMost artificial plasmas are generated by the application of electric and/or magnetic fields. Plasma generated in a laboratory setting and for industrial use can be generally categorized by:
Examples of industrial/commercial plasmaLow-pressure dischargesGlow discharge plasmas: Non-thermal plasmas generated by the application of DC or low frequency RF (<100 kHz) electric field to the gap between two metal electrodes. Probably the most common plasma; this is the type of plasma generated within fluorescent light tubes. Atmospheric pressureArc: This is a high power thermal discharge of very high temperature ~10,000 K. It can be generated using various power supplies. It is commonly used in metallurgical processes. For example it is used to melt rocks containing Al203 to produce Aluminum. Fields of active researchThis is just a partial list of topics. A more complete and organized list can be found on the Web site for Plasma science and technology.[22]
See also
|
|||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Plasma_(physics)". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||||||||||





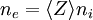 ), but on the scale of the Debye length there can be charge imbalance. In the special case that
), but on the scale of the Debye length there can be charge imbalance. In the special case that 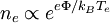 .
.
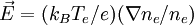 .
.


