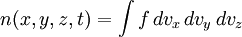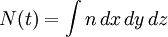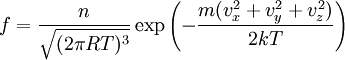To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Distribution function
In molecular kinetic theory in physics, a particle's distribution function is a function of seven variables, f(x,y,z,t;vx,vy,vz), which gives the number of particles per unit volume in phase space. It is the number of particles having approximately the velocity (vx,vy,vz) near the place (x,y,z) and time (t). The usual normalization of the distribution function is Product highlightHere, N is the total number of particles and n is the number density of particles - the number of particles per unit volume, or the density divided by the mass of individual particles. Particle distribution functions are often used in plasma physics to describe wave-particle interactions and velocity-space instabilities. Distribution functions are also used in fluid mechanics and statistical mechanics. The basic distribution function uses the Boltzmann constant k and temperature T with the number density to modify the normal distribution: Related distribution functions may allow bulk fluid flow, in which case the velocity origin is shifted, so that the exponent's numerator is m((vx − ux)2 + (vy − uy)2 + (vz − uz)2); (ux,uy,uz) is the bulk velocity of the fluid. Distribution functions may also feature non-isotropic temperatures, in which each term in the exponent is divided by a different temperature. Plasma theories such as magnetohydrodynamics may assume the particles to be in thermodynamic equilibrium. In this case, the distribution function is Maxwellian. This distribution function allows fluid flow and different temperatures in the directions parallel to, and perpendicular to, the local magnetic field. More complex distribution functions may also be used since plasmas are rarely in thermal equilibrium. The mathematical analog of a distribution is a measure; the time evolution of a measure on a phase space is the topic is study in dynamical systems. |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Distribution_function". A list of authors is available in Wikipedia. |