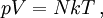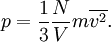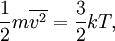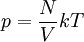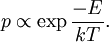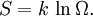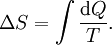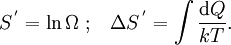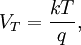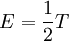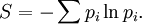To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Boltzmann constant
The Boltzmann constant (k or kB) is the physical constant relating temperature to energy. It is named after the Austrian physicist Ludwig Boltzmann, who made important contributions to the theory of statistical mechanics, in which this constant plays a crucial role. Product highlight
Bridge from macroscopic to microscopic physicsBoltzmann's constant k is a bridge between macroscopic and microscopic physics. Macroscopically, the ideal gas law states that, for an ideal gas, the product of pressure p and volume V is proportional to the product of amount of substance n (in number of moles) and absolute temperature T. where
Introducing Boltzmann's constant transforms this into an equation about the microscopic properties of molecules, where N is the number of molecules of gas, and k is Boltzmann's constant. Role in the equipartition of energyGiven a thermodynamic system at an absolute temperature T, the thermal energy carried by each microscopic "degree of freedom" in the system is on the order of magnitude of kT/2 (i.e., about 2.07×10−21 J, or 0.013 eV at room temperature). Application to simple gas thermodynamicsIn classical statistical mechanics, this average is predicted to hold exactly for homogeneous ideal gases. Monatomic ideal gases possess 3 degrees of freedom per atom, corresponding to the three spatial directions, which means a thermal energy of 1.5kT per atom. As indicated in the article on heat capacity, this corresponds very well with experimental data. The thermal energy can be used to calculate the root mean square speed of the atoms, which is inversely proportional to the square root of the atomic mass. The root mean square speeds found at room temperature accurately reflect this, ranging from 1370 m/s for helium, down to 240 m/s for xenon. Kinetic theory gives the average pressure p for an ideal gas as Substituting that the average translational kinetic energy is and gives so the ideal gas equation is regained. The ideal gas equation is also followed quite well for molecular gases; but the form for the heat capacity is more complicated, because the molecules possess new internal degrees of freedom, as well as the three degrees of freedom for movement of the molecule as a whole. Diatomic gases, for example, possess in total approximately 5 degrees of freedom per molecule. Role in Boltzmann factorsMore generally, systems in equilibrium with a reservoir of heat at temperature T have probabilities of occupying states with energy E weighted by the corresponding Boltzmann factor: Again, it is the energy-like quantity kT which takes central importance. Consequences of this include (in addition to the results for ideal gases above), for example the Arrhenius equation of simple chemical kinetics. Role in definition of entropyIn statistical mechanics, the entropy S of an isolated system at thermodynamic equilibrium is defined as the natural logarithm of Ω, the number of distinct microscopic states available to the system given the macroscopic constraints (such as a fixed total energy E): This equation, which relates the microscopic details of the system (via Ω) to its macroscopic state (via the entropy S), is the central idea of statistical mechanics. Such is its importance that it is inscribed on Boltzmann's tombstone. The constant of proportionality k appears in order to make the statistical mechanical entropy equal to the classical thermodynamic entropy of Clausius: One could choose instead a rescaled entropy in microscopic terms such that This is a rather more natural form; and this rescaled entropy exactly corresponds to Shannon's subsequent information entropy, and could thereby have avoided much unnecessary subsequent confusion between the two. Role in semiconductor physics: the thermal voltageIn semiconductors, the relationship between the flow of electrical current and the electrostatic potential across a p-n junction depends on a characteristic voltage called the thermal voltage, denoted VT. The thermal voltage depends on absolute temperature T (in kelvins) as where q is the magnitude of the electrical charge (in coulombs) on the electron (see elementary charge) with a value 1.602 176 487 ×10−19 C . Using the unit of electronvolt, the Boltzmann constant relating temperature to energy can be expressed as 8.617 343(15)×10−5 eV/K, making it easy to calculate that at room temperature (T ≈ 300 K), the value of the thermal voltage is approximately 25.85 millivolts ≈ 26 mV (Google calculator). See also semiconductor diodes. Boltzmann's constant in Planck unitsPlanck's system of natural units is one system constructed such that the Boltzmann constant is 1. This gives as the average kinetic energy of a gas molecule per degree of freedom; and makes the definition of thermodynamic entropy coincide with that of information entropy: The value chosen for the Planck unit of temperature is that corresponding to the energy of the Planck mass—a staggering 1.41679×1032 K. Historical noteAlthough Boltzmann first linked entropy and probability in 1877, it seems the relation was never expressed with a specific constant until Max Planck first introduced k , and gave an accurate value for it, in his derivation of the law of black body radiation in December 1900. The iconic terse form of the equation S = k log W on Boltzmann's tombstone is in fact due to Planck, not Boltzmann. As Planck wrote in his 1918 Nobel Prize lecture,
Before 1900, equations involving Boltzmann factors were not written using the energies per molecule and Boltzmann's constant, but rather using the gas constant R, and macroscopic energies for macroscopic quantities of the substance; as for convenience is still generally the case in Chemistry to this day. Value in different units
The digits in parentheses are the standard measurement uncertainty in the last two digits of the measured value. k can also be expressed with the unit mol (such as 1.99 calories/mole-kelvin), for historical reasons it is then called gas constant. The numerical value of k has no particular fundamental significance in itself: It merely reflects a preference for measuring temperature in units of familiar kelvins, based on the macroscopic physical properties of water. What is physically fundamental is the characteristic energy kT at a particular temperature. The numerical value of k measures the conversion factor for mapping from this characteristic microscopic energy E to the macroscopically-derived temperature scale T = E/k . If, instead of talking of room temperature as 300 K (27 °C or 80 °F), it were conventional to speak of the corresponding energy kT of 4.14×10−21 J, or 0.0259 eV, then Boltzmann's constant would not be needed. References
Categories: Statistical mechanics | Thermodynamics |
||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Boltzmann_constant". A list of authors is available in Wikipedia. | ||||||||||||||||||||||





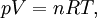
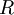 is called the
is called the