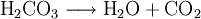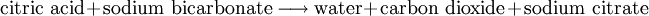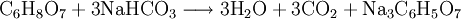To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Carbonation
Carbonation occurs when carbon dioxide is dissolved in water or an aqueous solution. This process yields the "fizz" to carbonated water and sparkling mineral water, the head to beer, and the cork pop and bubbles to champagne and sparkling wine. It is also behind the Diet Coke and Mentos eruption effect. Product highlight
EffervescenceLook up effervescence in Wiktionary, the free dictionary.
Effervescence is the escape of gas from an aqueous solution. The term is used to describe the foaming or fizzing that results from a release of gas. In the lab, a common example of effervescence is the addition of hydrochloric acid to a block of limestone. If a few pieces of marble or an antacid tablet are put in hydrochloric acid in a test tube fitted with a cork, effervescence of carbon dioxide can be witnessed. This process is generally represented by the following reaction, where a pressurized dilute solution of carbonic acid in water releases gaseous carbon dioxide at decompression:
In simple terms, it is the result of the chemical reaction occurring in the liquid which produces a gaseous product. Fizz"Fizz" is a word that is used to describe the action or sound of gas bubbles moving through and escaping from a liquid. Fizz also describes the formation of a foam of this gas and liquid at the top of the liquid's container. The word itself is an example of onomatopoeia, derived from the sound the multiple bubbles make together as they "pop" when they escape. A carbonated beverage, such as cola or beer, will form bubbles when the dissolved carbon dioxide is depressurized to form emulsions at the top, and it will make "fizzing" sounds when it is opened or poured into a container. In the United Kingdom, soft drinks are often referred to as 'fizzy drinks'. A cocktail based on carbonated water and an acidic juice is called a Fizz, such as the Gin Fizz. Fizz can also result from a chemical reaction, such as a solid dissolving in a liquid to produce gas. For example, Alka-Seltzer brand tablets, used to treat stomach indigestion, form an effervescent solution that fizzes when dropped into water. The essential chemical reaction is:
ShakingIt is commonly said that shaking a carbonated beverage will cause large amounts of foam to erupt upon opening, and it is often believed that shaking a bottle containing a carbonated beverage will cause the pressure inside to rise. In fact, when a pressure gauge is attached to a pressurized bottle of a carbonated beverage, it is found that the pressure within does not increase. It is instead the formation of tiny bubbles from the agitation that causes the foam; upon opening, the size of the bubbles will rapidly increase due to the reduction in pressure, resulting in excessive foaming. Agitating carbonated liquid in a resealed vessel increases the rate that CO2 is released from the solution and the rate that it approaches equilibrium pressure. The equilibrium pressure depends on the temperature, composition, and purity of the liquid and is not affected by agitation.
Natural and artificial carbonationCarbonation can occur as a result of natural processes: when yeast ferments dissolved sugars sealed in a pressure-tolerant bottle or keg; when underground volcanic carbon dioxide carbonates well water; or when rainwater passes through limestone into a cave and forms a stalactite. Or it can be done artificially by dissolving carbon dioxide under pressure into the liquid. Sometimes natural carbonation is called conditioning while the term carbonation is reserved for the artificial process. UsesIn many consumer beverages such as soft drinks (well known examples include Coca-Cola, 7 Up and Pepsi), carbonation is used to give "bite". Contrary to popular belief, the fizzy taste is caused by dilute carbonic acid inducing a slight burning sensation, and is not caused by the presence of bubbles. This can be shown by drinking a fizzy drink in a hyperbaric chamber at the same pressure as the beverage. This gives much the same taste, but the bubbles are completely absent. Carbonation is sometimes used for reasons other than consumption, to lower the pH (raise the hydrogen ion concentration) of a water solution, for example. BrewingIn homebrewing, overcarbonation can be dangerous; it can result in bottles gushing or even exploding. Adding priming sugar or malt extract at bottling time to beer that has had its fermentable sugar content totally consumed is the safest approach to carbonation. Exceeding recommended levels of priming sugar for a given recipe is dangerous, as is using inappropriate bottles or improper capping methods. Beer may also be force-carbonated using a keg and special bottling equipment so that the carbonation level can be carefully controlled.
Further reading
ReferencesSee also
|
|||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Carbonation". A list of authors is available in Wikipedia. |