To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Iron oreIron ores are rocks and minerals from which metallic iron can be economically extracted. The ores are usually rich in iron oxides and vary in colour from dark grey, bright yellow, deep purple, to rusty red. The iron itself is usually found in the form of magnetite (Fe3O4), hematite (Fe2O3), goethite, limonite or siderite. Hematite is also known as "natural ore". The name refers to the early years of mining, when certain hematite ores contained 66% iron and could be fed directly into iron making blast furnaces. Iron ore is the raw material used to make pig iron, which is one of the main raw materials to make steel. 98% of the mined iron ore is used to make steel.[1] Product highlight
Mining
World consumption of iron ore grows 10% per annum on average with the main consumers being China, Japan, Korea, the United States and the European Union. Iron ore mining methods vary by the type of ore being mined. There are four main types of iron ore deposits worked currently, depending on the mineralogy and geology of the ore deposits. These are magnetite, titanomagnetite, massive hematite and pisolitic ironstone deposits. Magnetite banded iron depositsBanded iron formations (BIF) are fine grained metamorphosed sedimentary rocks composed predominantly of magnetite and silica (as quartz). Banded Iron formations are locally known as taconite within North America. Mining of BIF formations involves coarse crushing and screening, followed by rough crushing and fine grinding to comminute the ore to the point where the crystallised magnetite and quartz are fine enough that the quartz is left behind when the resultant powder is passed under a magnetic separator. The key economic parameters for magnetite ore being economic are the crystallinity of the magnetite, the grade of the iron within the BIF host rock, and the contaminant elements which exist within the magnetite concentrate. The size and strip ratio of most magnetite resources is irrelevant as BIF formations can be hundreds of metres thick, with hundreds of kilometres of strike, and can easily come to more than 2,500 million tonnes of contained ore. The typical grade of iron at which a magnetite-bearing banded iron formation becomes economic is roughly 25% Fe, which can generally yield a 33% to 40% recovery of magnetite by weight, to produce a concentrate grading in excess of 64% Fe by weight. The typical magnetite iron ore concentrate has less than 0.1% phosphorus, 3-7% silica and less than 3% aluminium. The grain size of the magnetite and its degree of comingling with the silica groundmass determine the grind size to which the rock must be comminuted to enable efficient magnetic separation to provide a high purity magnetite concentrate. This determines the energy inputs required to run a milling operation. Generally most magnetite BIF deposits must be ground to between 32 and 45 micrometres in order to provide a low-silica magnetite concentrate. Magnetite concentrate grades are generally in excess of 63% Fe by weight and usually are low phosphorus, low aluminium, low titanium and low silica and demand a premium price. Currently magnetite iron ore is mined in Minnesota and Michigan in the U.S., and Eastern Canada mine taconite. Magnetite bearing BIF is currently mined extensively in Brazil, which exports significant quantities to Asia, and there is a nascent and large magnetite iron ore industry in Australia. Magmatic magnetite ore depositsOccasionally granite and ultrapotassic igneous rocks segregate magnetite crystals and form masses of magnetite suitable for economic concentration. A few iron ore deposits, notably in Chile, are formed from volcanic flows containing significant accumulations of magnetite phenocrysts. Chilean magnetite iron ore deposits within the Atacama Desert have also formed alluvial accumulations of magnetite in streams leading from these volcanic formations. Some magnetite skarn and hydrothermal deposits have been worked in the past as high-grade iron ore deposits requiring little beneficiation. There are several granite-associated deposits of this nature in Malaysia and Indonesia. Other sources of magnetite iron ore include metamorphic accumulations of massive magnetite ore such as at Savage River, Tasmania, formed by shearing of ophiolite ultramafics. Another, minor, source of iron ores are magmatic accumulations in ultramafic to mafic layered intrusions which contain a typically titanium-bearing magnetite crystal rock (magnetitite) often with vanadium. These ores form a niche market, with specialty smelters used to recover the iron, titanium and vanadium. These ores are beneficiated essentially similar to banded iron formation ores, but usually are more easily upgraded via crushing and screening. The typical titanomagnetite concentrate grades 57% Fe, 12% Ti and 0.5% V2O5. Hematite oreHematite iron ore deposits are currently exploited on all continents, with the largest intensity of exploitation in South America, Australia and Asia. Most large hematite iron ore deposits are sourced from metasomatically altered banded iron formations and rarely igneous accumulations. Hematite iron is typically rarer than magnetite bearing BIF or other rocks which form its main source or protolith rock, but it is considerably cheaper and easier to beneficiate the hematite ores and requires considerably less energy to crush and grind. Hematite ores however can contain significantly higher concentrations of penalty elements, typically being higher in phosphorus, water content (especially pisolite sedimentary accumulations) and aluminium (clays within pisolites). In Australia iron ore is won from three main sources: pisolite "channel iron deposit" ore derived by mechanical erosion of primary banded-iron formations and accumulated in alluvial channels such as at Pannawonica, Western Australia; and the dominant metasomatically-altered banded iron formation related ores such as at Newman, the Chichester Range, the Hamersley Range and Koolyanobbing, Western Australia. Other types of ore are coming to the fore recently, such as oxidised ferruginous hardcaps, for instance laterite iron ore deposits near Lake Argyle in Western Australia. The total recoverable reserves of iron ore in India are about 9,602 million tones of hematite and 3,408 million tones of magnetite. Madhya Pradesh, Karnataka, Bihar, Orissa, Goa, Maharashtra, Andhra Pradesh, Kerala, Rajasthan and Tamil Nadu are the principal Indian producers of iron ore. Consumption and economicsIron is the world's most commonly used metal. It is used primarily in structural engineering applications and in maritime purposes, automobiles, and general industrial applications (machinery). Iron-rich rocks are common worldwide, but ore-grade commercial mining operations are dominated by the countries listed in the table aside. The major constraint to economics for iron ore deposits is not necessarily the grade or size of the deposits, because it is not particularly hard to geologically prove enough tonnage of the rocks exist. The main constraint is the position of the iron ore relative to market, the cost of rail infrastructure to get it to market and the energy cost required to do so. World production averages one billion metric tons of raw ore annually. The world's largest producer of iron ore is the Brazilian mining corporation CVRD, followed by Australian company BHP Billiton and the Anglo-Australian Rio Tinto Group. A further Australian supplier, Fortescue Metals Group Ltd, is currently entering the development stage and may eventually bring Australia's production to second in the world. China is currently the largest consumer of iron ore, which translates to be the world's largest steel producing country. China is followed by Japan and Korea, which consume a significant amount of raw iron ore and metallurgical coal. In 2006, China produced 588 million tons of iron ore, with an annual growth of 38%. Pure iron is virtually unknown on the surface of the Earth except as Fe-Ni alloys from meteorites and very rare forms of deep mantle xenoliths. Therefore, all sources of iron used by human industry exploit iron oxide minerals, the primary form which is used in industry being hematite. However, in some situations, more inferior iron ore sources have been used by industrialized societies when access to high-grade hematite ore was not available. This has included utilisation of taconite in the United States, particularly during World War II, and goethite or bog ore used during the American Revolution and the Napoleonic wars. Magnetite is often used because it is magnetic and hence easily liberated from the gangue minerals. Inferior sources of iron ore generally required beneficiation. Due to the high density of hematite relative to silicates, beneficiation usually involves a combination of crushing and milling as well as heavy liquid separation. This is achieved by passing the finely crushed ore over a bath of solution containing bentonite or other agent which increases the density of the solution. When the density of the solution is properly calibrated, the hematite will sink and the silicate mineral fragments will float and can be removed. Taconite mining involves moving tremendous amounts of ore and waste. The waste comes in two forms, bedrock in the mine (mullock) that isn't ore, and unwanted minerals which are an intrinsic part of the ore rock itself (gangue). The mullock is mined and piled in waste dumps, and the gangue is separated during the beneficiation process and is removed as tailings. Taconite tailings are mostly the mineral quartz, which is chemically inert. This material is stored in large, regulated water settling ponds. Magnetite is beneficiated by crushing and then separating the magnetite from the gangue minerals with a magnet. This is usually so efficient that lower grade ore can be treated when it is magnetite than a comparable grade of hematite ore, especially when the magnetite is quite coarse. To convert an oxide of iron to metallic iron it must be smelted or sent through a direct reduction process.
DepletionIron ore reserves at present seem quite vast, but some are starting to suggest that the maths of continual exponential increase in consumption can even make this resource seem quite finite. For instance, Lester Brown of the Worldwatch Institute has suggested iron ore could run out within 64 years based on an extremely conservative extrapolation of 2% growth per year.[3] SmeltingIron ore consists of oxygen and iron atoms bonded together into molecules. To create pure iron, the ore must be smelted to remove the oxygen. Oxygen-iron bonds are strong, and to remove the iron from the oxygen, a stronger elemental bond must be presented to attach to the oxygen. Carbon is used because the strength of a carbon-oxygen bond is greater than that of the iron-oxygen bond, at high temperatures. Thus, the iron ore must be powdered and mixed with coke, to be burnt in the smelting process. However, this is not entirely as simple as that; carbon monoxide is the primary ingredient of chemically stripping oxygen from iron. Thus, the iron and carbon smelting must be kept at an oxygen deficient reduced state to promote burning of carbon to produce CO not CO2.
Trace Elements:Effects and RemediesThe inclusion of even small amounts of some elements can have profound effects on the behavioral characteristics of a batch of iron or the operation of a smelter. These effects can be both good and bad. Some catastrophically bad. Some chemicals were deliberately added. The addition of a flux made a blast furnace more efficient. Others were added because they made the iron more fluid, harder, or some other desirable quality. The choice of ore, fuel, and flux determined how the slag behaved and the operational characteristics of the iron produced. Ideally iron ore contains only iron and oxygen. In nature this is rarely the case. Typically, iron ore contains a host of elements which are often unwanted in modern steel. SiliconSilica (SiO2) is almost always present in iron ore. Most of it is slagged off during the smelting process. But, at temperatures above 1300°C some will be reduced and form an alloy with the iron. The hotter the furnace, the more silicon will be present in the iron. It is not uncommon to find up to 1.5% Si in European cast iron from the 16th to 18th centuries. The major effect of silicon is to promote the formation of gray iron. Gray iron is less brittle and easier to finish than white iron. It was preferred for casting purposes for this reason. Turner (1900:192-7) reported that silicon also reduced shrinkage and the formation of blowholes, lowering the number of bad castings. PhosphorusPhosphorus (P) has four major effects on iron: increased hardness and strength, lower solidus temperature, increased fluidity, and cold shortness. Depending on the use intended for the iron, these effects are either good or bad. Bog ore often has a high Phosphorus content (Gordon 1996:57). The strength and hardness of iron increases with the concentration of phosphorus. 0.05% phosphorus in wrought iron makes it as hard as medium carbon steel. High phosphorus iron can also be hardened by cold hammering. The hardening effect is true for any concentration of phosphorus. The more phosphorus, the harder the iron becomes and the more it can be hardened by hammering. Modern steel makers can increase hardness by as much as 30%, without sacrificing shock resistance by maintaining phosphorus levels between 0.07 and 0.12%. It also increases the depth of hardening due to quenching, but at the same time also decreases the solubility of carbon in iron at high temperatures. This would decrease its usefulness in making blister steel (cementation), where the speed and amount of carbon absorption is the overriding consideration. The addition of phosphorus has a down side. At concentrations higher than 0.2% iron becomes increasingly cold short, or brittle at low temperatures. Cold short is especially important for bar iron. Although, bar iron is usually worked hot, its uses often require it to be tough, bendable, and resistant to shock at room temperature. A nail that shattered when hit with a hammer or a carriage wheel that broke when it hit a rock would not sell well. High enough concentrations of phosphorus render any iron unuseable (Rostoker and Bronson 1990:22). The effects of cold shortness are magnified by temperature. Thus, a piece of iron that is perfectly serviceable in summer, might become extremely brittle in winter. There is some evidence that during the Middle Ages the very wealthy may have had a high phosphorus sword for summer and a low phosphorus sword for winter (Rostoker and Bronson 1990:22). Careful control of phosphorus can be of great benefit in casting operations. Phosphorus depresses the liquidus temperature, allowing the iron to remain molten for longer and increases fluidity. The addition of 1% can double the distance molten iron will flow (Rostoker and Bronson 1990:22). The maximum effect, about 500°C, is achieved at a concentration of 10.2% (Rostocker and Bronson 1990:194). For foundry work Turner felt the ideal iron had 0.2-0.55% phosphorus. The resulting iron filled molds with fewer voids and also shrank less. In the 19th century some producers of decorative cast iron used iron with up to 5% phosphorus. The extreme fluidity allowed them to make very complex and delicate castings. But, they could not be weight bearing, as they had no strength (Turner 1900:202-4). There are two remedies for high phosphorus iron. The oldest, and easiest, was avoidance. If the iron your ore produced was cold short, you found a new source of ore. The second method involves oxidizing the phosphorus during the fining process by adding iron oxide. The technique is usually associated with puddling in the 19th century, and may not have been understood earlier. For instance Isaac Zane, the owner of Marlboro Iron Works did not appear to know about it in 1772. Given Zane's reputation for keeping abreast of the latest developments, the technique was probably unknown to the ironmasters of Virginia and Pennsylvania. Phosphorus is a deleterious contaminant because it makes steel brittle, even at concentrations of as little as 0.5%. Phosphorus cannot be easily removed by fluxing or smelting, and so iron ores must generally be low in phosphorus to begin with. The iron pillar of India which does not rust is protected by a phosphoric composition. Phosphoric acid is used at a rust converter because phosphoric iron is less susceptible to oxidation. AluminiumSmall amounts of aluminium (Al) is present in many ores (often as clay) and some limestone. The former can be removed by washing the ore prior to smelting. Until the introduction of brick lined furnaces the amounts are small enough that they do not have an effect on either the iron or slag. However, when brick is used for hearths and the interior of blast furnaces, the amount of aluminium increases dramatically. This is due to the erosion of the furnace lining by the liquid slag, Aluminium is very hard to reduce. As a result aluminium contamination of the iron is not a problem. However, it does increase the viscosity of the slag (Kato and Minowa 1969:37 and Rosenqvist 1983:311). This will have a number of adverse effects on furnace operation. The thicker slag will slow the descent of the charge, prolonging the process. High aluminium will also make it more difficult to tap off the liquid slag. At the extreme this could lead to a frozen furnace. There are a number of solutions to a high aluminium slag. the first is avoidance, don't use ore or a lime source with a high aluminium content. Increasing the ratio of lime flux will decrease the viscosity (Rosenqvist 1983:311). SulfurSulfur (S) is a frequent contaminant in coal and coke. It was the presence of sulfur that prevented the use of coal in blast furnaces until 1709. Iron sulfide (pyrite, FeS2), is a common iron ore. It is also present in small quantities in many ores. Sulfur dissolves readily in both liquid and solid iron at the temperatures present in iron smelting. The effects of even small amounts of sulfur are immediate and serious. They were one of the first worked out by iron makers. Sulfur causes iron to be red or hot short (Gordon 1996:7). In modern operations, sulfur is unwanted because it produces undesirable sulfur dioxide gases in the flue emissions from a smelter and interferes with the smelting process. Hot short iron is brittle when hot. This was a serious problem as most iron used during the 17th and 18th century was bar or wrought iron. Wrought iron is shaped by repeated blows with a hammer while hot. A piece of hot short iron will crack if worked with a hammer. When a piece of hot iron or steel cracks the exposed surface immediately oxidizes. This layer of oxide prevents the mending of the crack by welding. Large cracks cause the iron or steel to break up. Smaller cracks can cause the object to fail during use. The degree of hot shortness is in direct proportion to the amount of sulfur present. Today iron with over 0.03% sulfur is avoided. Hot short iron can be worked, but it has to be worked at low temperatures. Working at lower temperatures requires more physical effort from the smith or forgeman. the metal must be struck more often and harder to achieve the same result. A mildly sulfur contaminated bar could be worked, but it required a great deal more time and effort. In cast iron sulfur promotes the formation of white iron. As little as 0.5% can counteract the effects of slow cooling and a high silicon content (Rostoker and Bronson 1990:21). White cast iron is more brittle, but also harder. It was generally avoided, because it was difficult to work. Except in China where high sulfur cast iron, some as high as 0.57%, made with coal and coke, was used to make bells and chimes (Rostoker, Bronson, and Dvorak 1984:760).According to Turner (1900:200), good foundry iron should have less than 0.15% sulfur. In the rest of the world a high sulfur cast iron could be used for making castings, but would make poor wrought iron. There are a number of remedies for sulfur contamination. The first, and the one most used in historic and prehistoric operations, was avoidance. Coal was not used in Europe (it was used in China) as a fuel for smelting because it contained sulfur and caused hot short iron. If an ore resulted in hot short metal, ironmasters found another ore. Sulfur can be removed from ores by roasting and washing. Roasting oxidizes sulfur to form sulfur dioxide which either escapes into the atmosphere or can be washed out. In warm climates it was possible to leave pyritic ore out in the rain. The combined action of rain, bacteria, and heat oxidize the sulfides to sulfates, which are water soluble (Turner 1900:77). Natural weathering was also used in Sweden. The same process, at geological speed, results in the gossan limonite ores. The importance attached to low sulfur iron is demonstrated by the consistently higher prices paid for the iron of Sweden, Russia, and Spain from the 16th to 18th centuries. Today sulfur is no longer a problem. The modern remedy is the addition of manganese. But, the operator must know how much sulfur is in the iron because at least five times as much manganese must be added to neutralize it. Some historic irons display manganese levels, but most are well below the level needed to neutralize sulfur (Rostoker and Bronson 1990:21). References
|
||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Iron_ore". A list of authors is available in Wikipedia. |





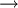 2CO.
2CO.


