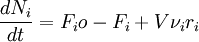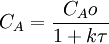To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Continuous stirred-tank reactor
The continuous stirred-tank reactor (CSTR), also known as vat- or backmix reactor, is a common ideal reactor type in chemical engineering. A CSTR often refers to a model is used to estimate the key unit operation variables when using a continuous agitated-tank reactor to reach a specified output. (See Chemical reactors.) The mathematical model works for all fluids: liquids, gases, and slurries. Product highlightAssume:
Integral mass balance on number of moles Ni of species i in a reactor of volume V. [accumulation] = [in] - [out] + [generation] 1. where Fio is the molar flow rate inlet of species i, Fi the molar flow rate outlet, and νi stoichiometric coefficient. The reaction rate, r, is generaly depentant on the reactant concentation and the rate constant (k). The rate constant can be figured by using the Arrhenius temperature dependence. Generally, as the temperature increases so does the rate at which the reaction occurs. Residence time, τ, is the average amount of time a discrete quantity of reagent spends inside the tank. Assume:
A → products NA = CA V (where CA is the concentration of species A, V is the volume of the reactor, NA is the number of moles of species A) 2. The values of the variables, outlet concentration and residence time, in Equation 2 are major design criteria. To model systems that do not obey the assumptions of constant temperature and a single reaction, additional dependent variables must be considered. If the system is considered to be in unsteady-state, a differential equation or a system of coupled differential equations must be solved. CSTR's are known to be one of the systems which exhibit complex behavior such as steady-state multiplicity, limit cycles and chaos. See also
References |
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Continuous_stirred-tank_reactor". A list of authors is available in Wikipedia. |