To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Double distilled waterDouble distilled water (abbreviated "ddH2O" or "Bidest. water") is prepared by double distillation of water. It is used, among other things, when single distillation does not lead to sufficiently pure water for some applications in biochemistry or trace analysis. Product highlightBidest water is used when pure, sterile water is essential. Whereas distilled water is enough for most chemical reactions, a molecular biologist attempting to create sterile, enzyme-free media might use bidest. water to ensure sterility. The theoretical pH of distilled water is 7.0. In practice, however, most distilled water will have a pH that is slightly acidic (less than 7.0) due to the presence of carbon dioxide (CO2) that is absorbed from the atmosphere. Dissolved carbon dioxide reacts slowly with water to give the bicarbonate and hydronium ions.
(carbonic acid, H2CO3, is only formed in strongly acid solutions). During distillation, the dissolved CO2 will be driven out of the liquid. However, during condensation the water will re-absorb the CO2 again resulting in a pH that is less than 7.0. See alsoCategories: Liquid water | Distillation |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Double_distilled_water". A list of authors is available in Wikipedia. |





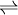 HCO3- + H3O+
HCO3- + H3O+


