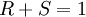To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Enantiomeric excessEnantiomeric excess exists where one enantiomer is present more than the other in a chemical substance. Product highlightSuch a mixture of two enantiomers, unlike a racemic mixture, will show a net optical rotation. It is possible to determine the specific rotation of the mixture and with knowledge of the specific rotation of the pure enantiomer the enantiomeric excess
The enantiomeric excess can be determined in another way if we know the amount of each enantiomer produced. If one knows the moles of each enantiomer produced then:
For example, in a sample with 40% ee in R, the remaining 60% is racemic with 30% of R and 30% of S, so that the total amount of R is 70%. Direct determination of these quantities is possible with NMR spectroscopy and chiral column chromatography. For mixtures of diastereomers the same treatment leads to diastereomeric excess. The term enantiomeric excess was introduced in 1971 by Morrison and Mosher in their publication Asymmetric Organic Reactions. Many practical observations challenge the supposed relationship and equality of equations 1 and 2:
The use of enantiomeric excess has established itself because of its historic ties with optical rotation. It has been suggested that the concept of ee should be replaced by that of er which stands for enantiomeric ratio or er (S:R) [2] or q (S/R) because determination of optical purity has been replaced by other techniques which directly measure R and S and because it simplifies many mathematical treatments. The same arguments are valid for changing diastereomeric excess (de) to diastereomeric ratio (dr). References
|
||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Enantiomeric_excess". A list of authors is available in Wikipedia. | ||||||||||





 can be determined.
can be determined.
![\ ee = ([\alpha]_{obs}/[\alpha]_{max})*100%](images/math/6/2/9/629a48618c0ab7dccf7d8c51d5dc5c82.png) (1)
(1)
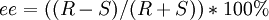 (2)
(2)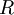 and
and 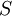 are the respective fractions of enantiomers in a mixture such that
are the respective fractions of enantiomers in a mixture such that