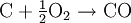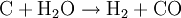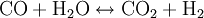To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
GasificationGasification is a process that converts carbonaceous materials, such as coal, petroleum, or biomass, into carbon monoxide and hydrogen by reacting the raw material at high temperatures with a controlled amount of oxygen. The resulting gas mixture is called synthesis gas or syngas and is itself a fuel. Gasification is a very efficient method for extracting energy from many different types of organic materials, and also has applications as a clean waste disposal technique. The advantage of gasification is that using the syngas is more efficient than direct combustion of the original fuel; more of the energy contained in the fuel is extracted. Syngas may be burned directly in internal combustion engines, used to produce methanol and hydrogen, or converted via the Fischer-Tropsch process into synthetic fuel. Gasification can also begin with materials that are not otherwise useful fuels, such as biomass or organic waste. In addition, the high-temperature combustion refines out corrosive ash elements such as chloride and potassium, allowing clean gas production from otherwise problematic fuels. Gasification of fossil fuels is currently widely used on industrial scales to generate electricity. However, almost any type of organic material can be used as the raw material for gasification, such as wood, biomass, or even plastic waste. Thus, gasification may be an important technology for renewable energy. In particular biomass gasification is carbon neutral. Gasification relies on chemical processes at elevated temperatures >700°C, which distinguishes it from biological processes such as anaerobic digestion that produce biogas. Product highlight
ChemistryIn a gasifier, the carbonaceous material undergoes several different processes:
In essence, a limited amount of oxygen or air is introduced into the reactor to allow some of the organic material to be "burned" to produce carbon monoxide and energy, which drives a second reaction that converts further organic material to hydrogen and additional carbon monoxide. HistoryThe gasification process was originally developed in the 1800s to produce town gas for lighting and cooking. Electricity and natural gas later replaced town gas for these applications, but the gasification process has been utilized for the production of synthetic chemicals and fuels since the 1920s. Wood gas generators, called Gasogene or Gazogène, were used to power motor vehicles in Europe during World War II fuel shortages.[1] Current applicationsIndustrial-scale gasification is currently mostly used to produce electricity from fossil fuels such as coal, where the syngas is burned in a gas turbine. Gasification is also used industrially in the production of electricity, ammonia and liquid fuels (oil) using Integrated Gasification Combined Cycles (IGCC), with the possibility of producing methane and hydrogen for fuel cells. IGCC is also a more efficient method of CO2 capture as compared to conventional technologies. IGCC demonstration plants have been operating since the early 1970s and some of the plants constructed in the 1990s are now entering commercial service. Within the last few years, gasification technologies have been developed that use plastic-rich waste as a feed. In a plant in Germany such a technology—on large scale—converts plastic waste via syngas into methanol.[2] Small-scale rural biomass gasifiers have been applied in India to a large extent, especially in the state of Tamil-Nadu in South India. Most of the applications are 9 kWe systems used for (drink) water pumping and street lighting operated by the local panchayat government. Although technically applicable the systems do face a number of problems. There are political, financial and maintenance problems. Most of the systems are no longer running after 1...3 years. Potential for renewable energyGasification can proceed from just about any organic material, including biomass and plastic waste. The resulting syngas burns cleanly into water vapor and carbon dioxide. Alternatively, syngas may be converted efficiently to methane via the Sabatier reaction, or diesel-like synthetic fuel via the Fischer-Tropsch process. Inorganic components of the input material, such as metals and minerals, are trapped in an inert and environmentally safe form as ash, which may have use as a fertilizer. Regardless of the final fuel form, gasification itself and subsequent processing neither emits nor traps greenhouse gasses such as carbon dioxide. Combustion of syngas or derived fuels does of course emit carbon dioxide. However, biomass gasification could play a significant role in a renewable energy economy, because biomass production removes CO2 from the atmosphere. While other biofuel technologies such as biogas and biodiesel are also carbon neutral, gasification runs on a wider variety of input materials, can be used to produce a wider variety of output fuels, and is an extremely efficient method of extracting energy from biomass. Biomass gasification is therefore one of the most technically and economically convincing energy possibilities for a carbon neutral economy [3] There is at present very little industrial scale biomass gasification being done. The Renewable Energy Network Austria[4] is associated with several successful biomass gasification demonstration projects, including a plant using dual fluidized bed gasification[5] that has supplied the town of Güssing with 2 MW of electricity and 4 MW of heat, generated from wood chips, since 2003. Gasification processesFour types of gasifier are currently available for commercial use: counter-current fixed bed, co-current fixed bed, fluidized bed and entrained flow.[6][7][8] The counter-current fixed bed ("up draft") gasifier consists of a fixed bed of carbonaceous fuel (e.g. coal or biomass) through which the "gasification agent" (steam, oxygen and/or air) flows in counter-current configuration. The ash is either removed dry or as a slag. The slagging gasifiers require a higher ratio of steam and oxygen to carbon in order to reach temperatures higher than the ash fusion temperature. The nature of the gasifier means that the fuel must have high mechanical strength and must be non-caking so that it will form a permeable bed, although recent developments have reduced these restrictions to some extent. The throughput for this type of gasifier is relatively low. Thermal efficiency is high as the gas exit temperatures are relatively low. However, this means that tar and methane production is significant at typical operation temperatures, so product gas must be extensively cleaned before use or recycled to the reactor. The co-current fixed bed ("down draft") gasifier is similar to the counter-current type, but the gasification agent gas flows in co-current configuration with the fuel (downwards, hence the name "down draft gasifier"). Heat needs to be added to the upper part of the bed, either by combusting small amounts of the fuel or from external heat sources. The produced gas leaves the gasifier at a high temperature, and most of this heat is often transferred to the gasification agent added in the top of the bed, resulting in an energy efficiency on level with the counter-current type. Since all tars must pass through a hot bed of char in this configuration, tar levels are much lower than the counter-current type. In the fluidized bed gasifier, the fuel is fluidized in oxygen and steam or air. The ash is removed dry or as heavy agglomerates that defluidize. The temperatures are relatively low in dry ash gasifiers, so the fuel must be highly reactive; low-grade coals are particularly suitable. The agglomerating gasifiers have slightly higher temperatures, and are suitable for higher rank coals. Fuel throughput is higher than for the fixed bed, but not as high as for the entrained flow gasifier. The conversion efficiency can be rather low due to elutriation of carbonaceous material. Recycle or subsequent combustion of solids can be used to increase conversion. Fluidized bed gasifiers are most useful for fuels that form highly corrosive ash that would damage the walls of slagging gasifiers. Biomass fuels generally contain high levels of corrosive ash. In the entrained flow gasifier a dry pulverized solid, an atomized liquid fuel or a fuel slurry is gasified with oxygen (much less frequent: air) in co-current flow. The gasification reactions take place in a dense cloud of very fine particles. Most coals are suitable for this type of gasifier because of the high operating temperatures and because the coal particles are well separated from one another. The high temperatures and pressures also mean that a higher throughput can be achieved, however thermal efficiency is somewhat lower as the gas must be cooled before it can be cleaned with existing technology. The high temperatures also mean that tar and methane are not present in the product gas; however the oxygen requirement is higher than for the other types of gasifiers. All entrained flow gasifiers remove the major part of the ash as a slag as the operating temperature is well above the ash fusion temperature. A smaller fraction of the ash is produced either as a very fine dry fly ash or as a black colored fly ash slurry. Some fuels, in particular certain types of biomasses, can form slag that is corrosive for ceramic inner walls that serve to protect the gasifier outer wall. However some entrained bed type of gasifiers do not possess a ceramic inner wall but have an inner water or steam cooled wall covered with partially solidified slag. These types of gasifiers do not suffer from corrosive slags. Some fuels have ashes with very high ash fusion temperatures. In this case mostly limestone is mixed with the fuel prior to gasification. Addition of a little limestone will usually suffice for the lowering the fusion temperatures. The fuel particles must be much smaller than for other types of gasifiers. This means the fuel must be pulverized, which requires somewhat more energy than for the other types of gasifiers. By far the most energy consumption related to entrained bed gasification is not the milling of the fuel but the production of oxygen used for the gasification. Waste disposalSeveral gasification processes for thermal treatment of waste are under development as an alternative to incineration. Waste gasification has several principal advantages over incineration:
A major challenge for waste gasification technologies is to reach an acceptable (positive) gross electric efficiency. The high efficiency of converting syngas to electric power is counteracted by significant power consumption in the waste preprocessing, production of large amounts of pure oxygen (which is often used as gasification agent), and gas cleaning. Another challenge becoming apparent when implementing the processes in real life is to obtain high availabilities of the plants, so that for example it is not necessary to close down the plant every few months for cleaning the reactor. Several waste gasification processes have been proposed, but few have yet been built and tested, and only a handful have been implemented as plants processing real waste, and always in combination with fossil fuels[10]. One plant (in Chiba, Japan using the Thermoselect process[11]) have been processing industrial waste since year 2000, but has yet not documented positive net energy production from the process. See also
References
Categories: Chemical engineering | Thermal treatment | Waste treatment technology | Unit processes |
|||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Gasification". A list of authors is available in Wikipedia. |