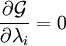To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Chemical equilibriumIn a chemical process, chemical equilibrium is the state in which the chemical activities or concentrations of the reactants and products have no net change over time. Usually, this would be the state that results when the forward chemical process proceeds at the same rate as their reverse reaction. The reaction rates of the forward and reverse reactions are generally not zero but, being equal, there are no net changes in any of the reactant or product concentrations. This process is known as dynamic equilibrium [1] [2]
Product highlight
IntroductionIn a chemical reaction, when reactants are mixed together in a reaction vessel (and heated if needed), the whole of reactants do not get converted into the products. After some time (which may be shorter than millionths of a second or longer than the age of the universe), there will come a point when a fixed amount of reactants will exist in harmony with a fixed amount of products, the amounts of neither changing anymore. This is called chemical equilibrium. The concept of chemical equilibrium was developed after Berthollet (1803) found that some chemical reactions are reversible. For any reaction such as to be at equilibrium the rates of the forward and backward (reverse) reactions have to be equal. In this chemical equation with harpoon arrows pointing both ways to indicate equilibrium, A and B are reactant chemical species, S and T are product species, and α, β, σ, and τ are the stoichiometric coefficients of the respective reactants and products. The equilibrium position of a reaction is said to lie far to the right if, at equilibrium, nearly all the reactants are used up and far to the left if hardly any product is formed from the reactants. Guldberg and Waage (1865), building on Berthollet’s ideas, proposed the law of mass action: where A, B, S and T are active masses and k+ and k− are rate constants. Since forward and backward rates are equal: and the ratio of the rate constants is also a constant, now known as an equilibrium constant. By convention the products form the numerator. Unfortunately, the law of mass action is valid only for concerted one-step reactions that proceed through a single transition state and is not valid in general because rate equations do not in general follow the stoichiometry of the reaction as Guldberg and Waage had proposed (see, for example, nucleophilic aliphatic substitution by SN1 or reaction of hydrogen and bromine to form hydrogen bromide). Equality of forward and backward reaction rates, however, is a necessary condition for chemical equilibrium, though it is not sufficient to explain why equilibrium occurs. Despite the failure of this derivation, the equilibrium constant for a reaction is indeed a constant, independent of the activities of the various species involved, though it does depend on temperature as observed by the van 't Hoff equation. Adding a catalyst will affect both the forward reaction and the reverse reaction in the same way and will not have an effect on the equilibrium constant. The catalyst will speed up both reactions thereby increasing the speed at which equilibrium is reached.[3] [4] Although the macroscopic equilibrium concentrations are constant in time reactions do occur at the molecular level. For example, in the case of ethanoic acid dissolved in water and forming ethanoate and hydronium ions,
a proton may hop from one molecule of ethanoic acid on to a water molecule and then on to an ethanoate ion to form another molecule of ethanoic acid and leaving the number of ethanoic acid molecules unchanged. This is an example of dynamic equilibrium. Equilibriums, like the rest of thermodynamics, are statistical phenomena, averages of microscopic behaviour. Le Chatelier's principle (1884) is a useful principle that gives a qualitative idea of an equilibrium system's response to changes in reaction conditions. If a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to counteract the change. For example, adding more S from the outside will cause an excess of products and the system will try to counteract this by increasing the reverse reaction and pushing the equilibrium point backward (though the equilibrium constant will stay the same). If mineral acid is added to the ethanoic acid mixture, increasing the concentration of hydronium ion, the amount of dissociation must decrease as the reaction is driven to the left in accordance with this principle. This can also be deduced from the equilibrium constant expression for the reaction: if {H3O+} increases {CH3CO2H} must increase and {CH3CO2−} must decrease. A quantitative version is given by the reaction quotient. J.W. Gibbs suggested in 1873 that equilibrium is attained when the Gibbs energy of the system is at its minimum value (assuming the reaction is carried out under constant pressure). What this means is, the derivative of the Gibbs energy with respect to reaction coordinate (a measure of the extent of reaction that has occurred, ranging from zero for all reactants to a maximum for all products) vanishes, signalling a stationary point. This derivative is usually called, for certain technical reasons, the Gibbs energy change.[5] This criterion is both necessary and sufficient. If a mixture is not at equilibrium the liberation of the excess Gibbs energy (or Helmholtz energy at constant volume reactions) is the “driving force” for the composition of the mixture to change until equilibrium is reached. The equilibrium constant can be related to the standard Gibbs energy change for the reaction by the equation where R is the universal gas constant and T the temperature. When the reactants are dissolved in a medium of high ionic strength the quotient of activity coefficients may be taken to be constant. In that case the concentration quotient, Kc, where [A] is the concentration of A, etc., is independent of the analytical concentration of the reactants. For this reason, equilibrium constants for solutions are usually determined in media of high ionic strength. Kc varies with ionic strength, temperature and pressure (or volume). Likewise Kp for gases depends on partial pressure. These constants are easier to measure and encountered in high-school chemistry courses. ThermodynamicsThe relationship between the Gibbs energy and the equilibrium constant can be found by considering chemical potentials. The thermodynamic condition for chemical equilibrium is[6]
In this article only the constant pressure case is considered. The constant volume case is important in geochemistry and atmospheric chemistry where pressure variations are significant. Note that if reactants and products were in standard state (completely pure) then there would be no reversibility and no equilibrium. The mixing of the products and reactants contributes a large entropy (known as entropy of mixing) to states containing equal mixture of products and reactants. The combination of the standard Gibbs energy change and the Gibbs energy of mixing determines the equilibrium state.[7] In general an equilibrium system is defined by writing an equilibrium equation for the reaction To meet the thermodynamic condition for equilibrium the Gibbs energy must be stationary, meaning that the derivative of G with respect to reaction coordinate (ΔG) must be zero. It can be shown that ΔG is in fact equal to the difference between the chemical potentials of the products and those of the reactants. Therefore, the sum of the Gibbs energies of the reactants must be the equal to the sum of the Gibbs energies of the products. where μ is in this case a partial molar Gibbs energy, a chemical potential. The chemical potential of a reagent A is a function of the activity, {A} of that reagent. Substituting expressions like this into the Gibbs energy equation: which at constant pressure and temperature becomes: results in: By substituting the chemical potentials: the relationship becomes: At equilibrium leading to: ΔGm Treatment of activityThe expression for the equilibrium constant can be re-written as the product of a concentration quotient, Kc and an activity coefficient quotient, Γ. [A] is the concentration of reagent A etc. It is possible in principle to obtain values of the activity coefficients, γ. For solutions equations such as the Debye-Hückel equation or extensions such as Davies equation[8] or Pitzer equations[9] may be used.Software (below). However this is not always possible. It is common practice to assume that Γ is a constant and to use the concentration quotient in place of the thermodynamic equilibrium constant. It is also general practice to use the term equilibrium constant instead of the more pedantically accurate concentration quotient. This practice will be followed here. For reactions in the gas phase partial pressure is used in place of concentration and fugacity coefficient in place of activity coefficient. In the real world, for example when making ammonia industrially, fugacity coefficients must be taken into account. Fugacity, f, is the product of partial pressure and fugacity coefficient. The chemical potential of a species in the gas phase is given by so the general expression defining an equilibrium constant is valid for both solution and gas phases. Justification for the use of concentration quotientsIn aqueous solution, equilibrium constants are usually determined in the presence of an "inert" electrolyte such as sodium nitrate NaNO3 or Potassium perchlorate KClO4. The ionic strength, I, of a solution containing a dissolved salt, X+Y-, is given by where c stands for concentration, z stands for ionic charge and the sum is taken over all the species in equilibrium. When the concentration of dissolved salt is much higher than the analytical concentrations of the reagents, the ionic strength is effectively constant. Since activity coefficients depend on ionic strength the activity coefficients of the species are effectively independent of concentration. Thus, the assumption that Γ is constant is justified. The concentration quotient is a simple multiple of the equilibrium constant.[10] However, Kc will vary with ionic strength. If it is measured at a series of different ionic strengths the value can be extrapolated to zero ionic strength.[9] The concentration quotient obtained in this manner is known, paradoxically, as a thermodynamic equilibrium constant. To use a published value of an equilibrium constant in conditions of ionic strength different from the conditions used in its determination, the value should be adjustedSoftware (below). Metastable mixturesA mixture may be appear to have no tendency to change, though it is not at equilibrium. For example, a mixture of SO2 and O2 is metastable as there is a kinetic barrier to formation of the product, SO3.
The barrier can be overcome when a catalyst is also present in the mixture as in the Contact process, but the catalyst does not affect the equilibrium concentrations. Likewise, the formation of bicarbonate from carbon dioxide and water is very slow under normal conditions
but almost instantaneous in the presence of the catalytic enzyme carbonic anhydrase. Pure compounds in equilibriaWhen pure substances (liquids or solids) are involved in equilibria they do not appear in the equilibrium equation [11] Applying the general formula for an equilibrium constant to the specific case of ethanoic acid one obtains It may be assumed that the concentration of water is constant. This assumption will be valid for all but very concentrated solutions. The equilibrium constant expression is therefore usually written as where now
a constant factor is incorporated into the equilibrium constant. A particular case is the self-ionization of water itself The self-ionization constant of water is defined as
It is perfectly legitimate to write [H+] for the hydronium ion concentration since the state of solvation of the proton is constant (in dilute solutions) and so does not affect the equilibrium concentrations. Kw varies with variation in ionic strength and/or temperature. The concentrations of H+ and OH- are not independent quantities. Most commonly [OH-] is replaced by Kw[H+]-1 in equilibrium constant expressions which would otherwise hydroxide. Solids also do not appear in the equilibrium equation. An example is the Boudouard reaction [11]: for which the equation (without solid carbon) is written as: Multiple equilibriaConsider the case of a dibasic acid H2A. When dissolved in water the mixture will contain H2A, HA- and A2-. This equilibrium can be split into two steps in each of which one proton is liberated. K1 and K2 are examples of stepwise equilibrium constants. The overall equilibrium constant,βD, is product of the stepwise constants. Note that these constants are dissociation constants because the products on the right hand side of the equilibrium expression are dissociation products. In many systems it is preferable to use association constants. β1 and β2 are examples of association constants. Clearly β1 = 1/K2 and β2 = 1/βD; lg β1 = pK2 and lg β2 = pK2 + pK1[12] Effect of temperature change on an equilibrium constantThe effect of changing temperature on an equilibrium constant is given by the van 't Hoff equation Thus, for exothermic reactions, (ΔH is negative) K decreases with temperature, but for endothermic reactions (ΔH is positive) K increases with temperature. An alternative formulation is At first sight this appears to offer a means of obtaining the standard molar enthalpy of the reaction by studying the variation of K with temperature. In practice, however, the method is unreliable because error propagation almost always gives very large errors on the values calculated in this way. Types of equilibrium and some applications
In these applications terms such as stability constant, formation constant, binding constant, affinity constant, association/dissociation constant are used. In biochemistry it is common to give units for binding constants, which serve to define the concentration units used when the constant’s value was determined. Composition of an equilibrium mixtureWhen the only equilibrium is that of the formation of a 1:1 adduct as the composition of a mixture, there are any number of ways that the composition of a mixture can be calculated. For example, see ICE table for a traditional method of calculating the pH of a solution of a weak acid. There are three approaches to the general calculation of the composition of a mixture at equilibrium.
Solving the equations of mass-balanceIn general the calculations are rather complicated. For instance, in the case of a dibasic acid, H2A dissolved in water the two reactants can be specified as the conjugate base, A2-, and the proton, H+. The following equations of mass-balance could apply equally well to a base such as 1,2-diaminoethane, in which case the base itself is designated as the reactant A: With TA the total concentration of species A. Note that it is customary to omit the ionic charges when writing and using these equations. When the equilibrium constants are known and the total concentrations are specified there are two equations in two unknown "free concentrations" [A] and [H]. This follows from the fact that [HA]= β1[A][H], [H2A]= β2[A][H]2 and [OH] = Kw[H]-1 so the concentrations of the "complexes" are calculated from the free concentrations and the equilibrium constants. General expressions applicable to all systems with two reagents, A and B would be It is easy to see how this can be extended to three or more reagents. Composition for polybasic acids as a function of pHThe composition of solutions containing reactants A and H is easy to calculate as a function of p[H]. When [H] is known the free concentration [A] is calculated from the mass-balance equation in A. Here is an example of the results that can be obtained.
This diagram, for the hydrolysis of the aluminum Lewis acid Al3+aq [15] shows the species concentrations for a 5×10-6M solution of an aluminium salt as a function of pH. Each concentration is shown as a percentage of the total aluminium. Solution equilibria with precipitationThe diagram above illustrates the point that a precipitate may be formed which is not one of the main species in the solution equilibrium. At pH just below 5.5 the main species present in a 5μM solution of Al3+ are aluminum hydroxides Al(OH)2+, Al(OH)2+ and Al13(OH)327+, but on raising the pH Al(OH)3 precipitates from the solution. This occurs because Al(OH)3 has a very large lattice energy. As the pH rises more and more Al(OH)3 comes out of solution. This is an example of Le Chatelier's principle in action: increasing the concentration of the hydroxide ion causes more aluminium hydroxide to precipitate, which removes hydroxide from the solution. When the hydroxide concentration becomes sufficiently high the soluble aluminate, Al(OH)4-, is formed. Another common instance where precipitation occurs is when a metal cation interacts with an anionic ligand to form an electrically neutral complex. If the complex is hydrophopbic it will precipitate out of water. This occurs with the nickel ion Ni2+ and dimethylglyoxime, (dmgH2): in this case the lattice energy of the solid is not particularly large, but it greatly exceeds the energy of solvation of the molecule Ni(dmgH)2. Minimization of Gibbs energyAt equilibrium, G is at a minimum: For a closed system, no particles may enter or leave, although they may combine in various ways. The total number of atoms of each element will remain constant. This means that the minimization above must be subjected to the constraints: where aij is the number of atoms of element i in molecule j and bi0 is the total number of atoms of element i, which is a constant, since the system is closed. If there are a total of k types of atoms in the system, then there will be k such equations. This is a standard problem in optimisation, known as constrained minimisation. The most common method of solving it is using the method of Lagrange multipliers, also known as undetermined multipliers (though other methods may be used). Define: where the λi are the Lagrange multipliers, one for each element. This allows each of the Nj to be treated independently, and it can be shown using the tools of multivariate calculus that the equilibrium condition is given by
(For proof see Lagrange multipliers) This is a set of (m+k) equations in (m+k) unknowns (the Nj and the λi) and may therefore be solved for the equilibrium concentrations Nj as long as the chemical potentials are known as functions of the concentrations at the given temperature and pressure. (See Thermodynamic databases for pure substances). This method of calculating equilibrium chemical concentrations is useful for systems with a large number of different molecules. The use of k atomic element conservation equations for the mass constraint is straightforward, and replaces the use of the stoichiometric coefficient equations. [13] See also
References
Further reading
Software for chemical equilibria
Categories: Analytical chemistry | Physical chemistry |
||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Chemical_equilibrium". A list of authors is available in Wikipedia. |





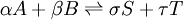
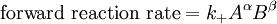
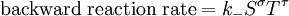
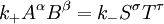
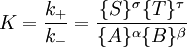
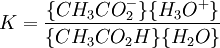
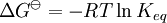
![K_c=\frac{[S]^\sigma [T]^\tau } {[A]^\alpha [B]^\beta}](images/math/a/f/6/af61c8307fbde1f9595004610ce90563.png)
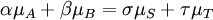
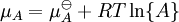
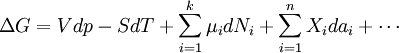
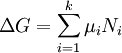
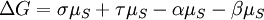
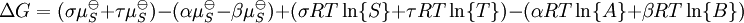
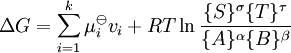
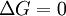 and therefore
and therefore
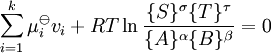
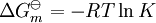
![K=\frac{{[S]} ^\sigma {[T]}^\tau ... } {{[A]}^\alpha {[B]}^\beta ...} \times \frac{{\gamma_S} ^\sigma {\gamma_T}^\tau ... } {{\gamma_A}^\alpha {\gamma_B}^\beta ...} = K_c \Gamma](images/math/e/a/3/ea34698e710811edf7462f812d3e2bcd.png)
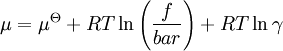
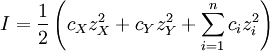
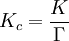
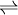 2SO3
2SO3
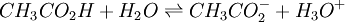
![K_c=\frac{[{CH_3CO_2}^-][{H_3O}^+]} {[{CH_3CO_2H}][{H_2O}]}](images/math/3/0/7/307fe3fe38fb84bace20e2b8ba8eeebd.png)
![K=\frac{[{CH_3CO_2}^-][{H_3O}^+]} {[{CH_3CO_2H}]}](images/math/5/7/d/57d1c1393a3f833fa1317baffe01d5d3.png)
![K=K_c*[H_2O]\,](images/math/2/0/8/208ece06e533814d5c022aa8cae90efe.png)
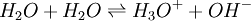
![K_w = [H^+][OH^-]\,](images/math/0/4/7/04751982906d78616e59229017e70bc8.png)
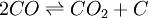
![K_c=\frac{[CO_2]} {[CO]^2}](images/math/6/d/3/6d35112295d85580245a93dbd6cfd011.png)
![H_2A \rightleftharpoons HA^- + H^+ :K_1=\frac{[HA^-][H^+]} {[H_2A]}](images/math/6/8/9/689ba8b4100e2ca021e92cc57c87829b.png)
![HA^- \rightleftharpoons A^{2-} + H^+ :K_2=\frac{[A^{2-}][H^+]} {[HA^-]}](images/math/5/0/8/508c0a5e1a5b68608b549a390a6c28f4.png)
![H_2A \rightleftharpoons A^{2-} + 2H^+ :\beta_D = \frac{[A^{2-}][H^+]^2} {[H_2A]}=K_1K_2](images/math/d/a/b/dabc558f3d7d75d148f9bea2e83f842e.png)
![A^{2-} + H^+ \rightleftharpoons HA^- :\beta_1=\frac {[HA^-]} {[A^{2-}][H^+]}](images/math/d/3/8/d387e931f865d551b3f7b992b1f638f3.png)
![A^{2-} + 2H^+ \rightleftharpoons H_2A :\beta_2=\frac {[H_2A]} {[A^{2-}][H^+]^2}](images/math/8/f/b/8fbf2ccc028a357fbf558f2c87b02c38.png)
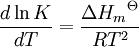
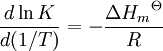
![T_A = [A] + [HA] +[H_2A] \,](images/math/b/a/a/baa30fecb1f077660b02981ea3e2165d.png)
![T_H = [H] + [HA] + 2[H_2A] - [OH] \,](images/math/a/f/0/af0c187ac39ffc5a18d3091fa7d4579d.png)
![T_A = [A] + \beta_1[A][H] + \beta_2[A][H]^2 \,](images/math/2/7/6/27647add7a20346a578e0a31316af783.png)
![T_H = [H] + \beta_1[A][H] + 2\beta_2[A][H]^2 - K_w[H]^{-1} \,](images/math/4/3/f/43fc06f15ea67771c7a47b911db13a31.png)
![T_A=[A]+\sum_i{p_i \beta_i[A]^{p_i}[B]^{q_i}}](images/math/a/6/6/a6688b731e5c97ba006bd88e5260263b.png)
![T_B=[B]+\sum_i{q_i \beta_i[A]^{p_i}[B]^{q_i}}](images/math/6/8/9/68978139960014c7dc00450e0d8e562e.png)
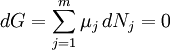
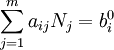
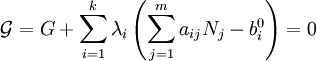
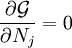 and
and