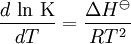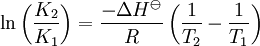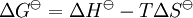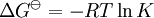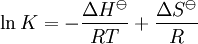To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Van 't Hoff equationThe van 't Hoff equation in chemical thermodynamics relates the change in temperature (T) to the change in the equilibrium constant (K) given the enthalpy change (ΔH). The equation was first derived by Jacobus Henricus van 't Hoff. Product highlightIf the enthalpy change of reaction is assumed to be constant with temperature, the definite integral of this differential equation between temperatures T1 and T2 is given by In this equation K1 is the equilibrium constant at absolute temperature T1 and K2 is the equilibrium constant at absolute temperature T2. ΔH Since and it follows that Therefore, a plot of the natural logarithm of the equilibrium constant versus the reciprocal temperature gives a straight line. The slope of the line is equal to minus the standard enthalpy change divided by the gas constant, ΔH |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Van_'t_Hoff_equation". A list of authors is available in Wikipedia. |