To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Reaction rate
The reaction rate or rate of reaction for a reactant or product in a particular reaction is intuitively defined as how fast a reaction takes place. For example, the oxidation of iron under the atmosphere is a slow reaction which can take many years, but the combustion of butane in a fire is a reaction that takes place in fractions of a second. Chemical kinetics is the part of physical chemistry that studies reaction rates. The concepts of chemical kinetics are applied in many disciplines, such as chemical engineering, enzymology and environmental engineering. Product highlight
Formal definition of reaction rateConsider a typical chemical reaction:
The lowercase letters (a, b, p, and q) represent stoichiometric coefficients, while the capital letters represent the reactants (A and B) and the products (P and Q). According to IUPAC's Gold Book definition[1] the reaction rate v (also r or R) for a chemical reaction occurring in a closed system under constant-volume conditions, without a build-up of reaction intermediates, is defined as: The IUPAC[1] recommends that the unit of time should always be the second. In such a case the rate of reaction differs from the rate of increase of concentration of a product P by a constant factor (the reciprocal of its stoichiometric number) and for a reactant A by minus the reciprocal of the stoichiometric number. Reaction rate usually has the units of mol dm-3 s-1. It is important to bear in mind that the previous definition is only valid for a single reaction, in a closed system of constant volume. This most usually implicit assumption must be stated explicitly, otherwise the definition is incorrect: If water is added to a pot containing salty water, the concentration of salt decreases, although there is no chemical reaction. For any system in general the full mass balance must be taken into account: IN - OUT + GENERATION = ACCUMULATION When applied to the simple case stated previously this equation reduces to: For a single reaction in a closed system of varying volume the so called rate of conversion can be is used, in order to avoid handling concentrations. It is defined as the derivative of the extent of reaction with respect to time.
When side products or reaction intermediates are formed, the IUPAC[1] recommends the use of the terms rate of appearance and rate of disappearance for products and reactants, respectively. Reaction rates may also be defined on a basis that is not the volume of the reactor. When a catalyst is used the reaction rate may be stated on a catalyst weight (mol g-1 s-1) or surface area (mol m-2 s-1) basis. If the basis is a specific catalyst site that may be rigorously counted by a specified method, the rate is given in units of s-1 and is called a turnover frequency. Factors influencing rate of reactionFactors that affect the rate of reaction:
For example, coal burns in a fireplace in the presence of oxygen but it doesn't when it is stored at room temperature. The reaction is spontaneous at low and high temperatures but at room temperature its rate is so slow that it is negligible. The increase in temperature, as created by a match, allows the reaction to start and then it heats itself, because it is exothermic. That is valid for many other fuels, such as methane, butane, hydrogen...
For example when methane reacts with chlorine in the dark, the reaction rate is very slow. It can be sped up when the mixture is put under diffused light. In bright sunlight, the reaction is explosive.
For example, platinum catalyzes the combustion of hydrogen with oxygen at room temperature.
All the factors that affect a reaction rate are taken into account in the rate equation of the reaction. Rate EquationFor a chemical reaction n A + m B → C + D, the rate equation or rate law is a mathematical expression used in chemical kinetics to link the rate of a reaction to the concentration of each reactant. It is of the kind: In this equation k(T) is the reaction rate coefficient or rate constant, although it is not really a constant, because it includes all the parameters that affect reaction rate, except for concentration, which is explicitly taken into account. Of all the parameters described before, temperature is normally the most important one. The exponents n' and m' are called reaction orders and depend on the reaction mechanism. They sometimes are the same as the stoichiometric coefficients of A and B, but not necessarily. Stoichiometry, molecularity (the actual number of molecules colliding) and reaction order only coincide necessarily in elementary reactions, that is, those reactions that take place in just one step. The reaction equation for elementary reactions coincides with the process taking place at the atomic level, i.e. n molecules of type A are colliding with m molecules of type B (n plus m is the molecularity). For gases the rate law can also be expressed in pressure units using e.g. the ideal gas law. By combining the rate law with a mass balance for the system in which the reaction occurs, an expression for the rate of change in concentration can be derived. For a closed system with constant volume such an expression can look like Temperature dependenceEach reaction rate coefficient k has a temperature dependency, which is usually given by the Arrhenius equation: Ea is the activation energy and R is the gas constant. Since at temperature T the molecules have energies given by a Boltzmann distribution, one can expect the number of collisions with energy greater than Ea to be proportional to The values for A and Ea are dependent on the reaction. There are also more complex equations possible, which describe temperature dependence of other rate constants which do not follow this pattern. Pressure dependenceThe pressure dependence of the rate constant for condensed-phase reactions (i.e., when reactants and products are solids or liquid) is usually suffficiently weak in the range of pressures normally encountered in industry that it is neglected in practice. The pressure dependence of the rate constant is associated with the activation volume. For the reaction proceeding through an activation-state complex: the activation volume, where For the above reaction, one can expect the change of the reaction rate constant (based either on mole-fraction or molal-concentration) with pressure at constant temperature to be: In practice, the matter can be complicated because the partial molar volumes and the activation volume can themselves be a function of pressure. Reactions can increase or decrease their rates with pressure, depeding on the value of ExamplesFor the reaction The rate equation is: The rate equation does not simply reflect the reactants stoichiometric coefficients in the overall reaction: it is first order in H2, although the stoichiometric coefficient is 2 and it is second order in NO. In chemical kinetics the overall reaction is usually proposed to occur through a number of elementary steps. Not all of these steps affect the rate of reaction; normally it is only the slowest elementary step that affect the reation rate. For example, in:
Reactions 1 and 3 are very rapid compared to the second, so it is the slowest reaction that is reflected in the rate equation. The slow step is considered the rate determining step. The orders of the rate equation are those from the rate determining step. See also
Notes
Categories: Chemical kinetics | Chemical engineering |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Reaction_rate". A list of authors is available in Wikipedia. |





![v = - \frac{1}{a} \frac{d[A]}{dt} = - \frac{1}{b} \frac{d[B]}{dt} = \frac{1}{p} \frac{d[P]}{dt} = \frac{1}{q} \frac{d[Q]}{dt}](images/math/0/1/9/019d4954e10b404cec525f8e8c662016.png)
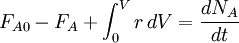
![v= \frac{d[A]}{dt}](images/math/a/7/d/a7d27a014710fc386f481d4ba8cc50c6.png)
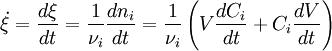
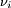 is the stoichiometric coefficient for substance i,
is the stoichiometric coefficient for substance i, 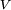 is the volume of reaction and
is the volume of reaction and 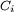 is the concentration of substance i.
is the concentration of substance i.
![\,r = k(T)[A]^{n'}[B]^{m'}](images/math/4/9/3/493ba8a5537c9100593d9360d9a59543.png)
![\frac{d[C]}{dt} = k(T)[A]^{n'}[B]^{m'}](images/math/7/3/e/73e873ab1b9e6162a0c45659b62d0b52.png)
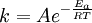
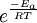 . A is the pre-exponential factor or frequency factor.
. A is the pre-exponential factor or frequency factor.
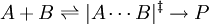
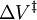 , is:
, is:
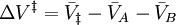
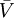 denote the partial molar volumes of the reactants and products, and
denote the partial molar volumes of the reactants and products, and 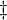 indicates the activation-state complex.
indicates the activation-state complex.
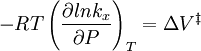
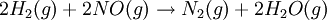
![r = k [H_2]^1[NO]^2 \,](images/math/9/4/7/94762b015683b6e69622261518e9e6da.png)
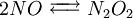 (fast equilibrium)
(fast equilibrium)
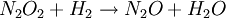 (slow)
(slow)
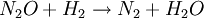 (fast)
(fast)


