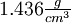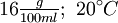To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Guanidine nitrateGuanidine nitrate is a high energy fuel used in some gas generator and solid rocket propellant applications. Product highlightOverviewGuanidine nitrate has chemical formula C(NH2)3NO3. It has been used as a monopropellant in the Jetex engine for model airplanes. It is attractive because it has a high gas output, low flame temperature, and non-toxic combustion products. It has a relatively high monopropellant specific impulse of 177 seconds (
References and External Links
Categories: Nitrates | Rocket fuels |
||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Guanidine_nitrate". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||





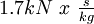 ).
).