To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Kramers-Wannier dualityThe Kramers-Wannier duality is a symmetry in statistical physics. It relates the free energy of a two-dimensional square-lattice Ising model at a low temperature to that of another Ising model at a high temperature. It was discovered by Hendrik Kramers and Gregory Wannier in 1941. With the aid of this duality Kramers and Wannier found the exact location of the critical point for the Ising model on the square lattice. Product highlightSimilar dualities establish relations between free energies of other statistical models. For instance, in 3 dimensions the Ising model is dual to an Ising gauge model. DerivationThe low temperature expansion for (K * ,L * ) is which by using the transformation gives where v = tanhK and w = tanhL, yielding a relation with the high-temperature expansion. The relations can written more symmetrically as
With the free energy per site in the thermodynamic limit the Kramers-Wannier duality gives In the isotropic case where K = L, if there is a critical point at K = Kc, then there is another at See alsoReferences
Categories: Statistical mechanics | Lattice models |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Kramers-Wannier_duality". A list of authors is available in Wikipedia. |





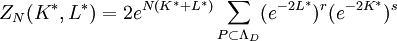
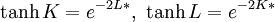
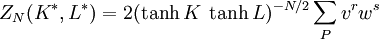
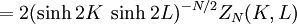
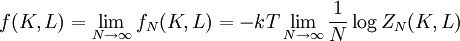
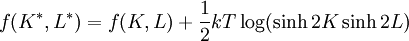
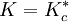 . Hence, in the case of there being a unique critical point, it would be located at
. Hence, in the case of there being a unique critical point, it would be located at 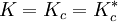 , implying
, implying 

