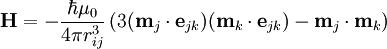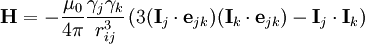To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Magnetic dipole-dipole interactionMagnetic dipole-dipole interaction, also called dipolar coupling, refers to the direct interaction between two magnetic dipoles. The energy of the interaction is as follows: Product highlightwhere ejk is a unit vector parallel to the line joining the centers of the two dipoles. rij is the distance between two dipoles, mk and mj. For two interacting nuclear spins: γj, γk and rij are gyromagnetic ratios of two spins and spin-spin distance respectively. Dipolar coupling and NMR spectroscopyThe direct dipole-dipole coupling is very useful for molecular structural studies, since it depends only on known physical constants and the inverse cube of internuclear distance. Estimation of this coupling provides a direct spectroscopic route to the distance between nuclei and hence the geometrical form of the molecule. Although internuclear magnetic dipole couplings contain a great deal of structural information, in isotropic solution, they average to zero as a result of rotational diffusion. However, their effect on nuclear spin relaxation results in measurable nuclear Overhauser effects (NOEs). The residual dipolar coupling (RDC) occur if the molecules in solution exhibit a partial alignment leading to an incomplete averaging of spatially anisotropic magnetic interactions i.e. dipolar couplings. RDC measurement provides information on the global folding of the protein-long distance structural information. It also provides information about "slow" dynamics in molecules.
References
See also
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Magnetic_dipole-dipole_interaction". A list of authors is available in Wikipedia. |