To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Mass-to-charge ratio
The mass-to-charge ratio, is a physical quantity that is widely used in the electrodynamics of charged particles, e.g. in electron optics and ion optics. It appears in the scientific fields of lithography, electron microscopy, cathode ray tubes, accelerator physics, nuclear physics, auger spectroscopy, cosmology and mass spectrometry. The importance of the mass-to-charge ratio, according to classical electrodynamics, is that two particles with the same mass-to-charge ratio move in the same path in a vacuum when subjected to the same electric and magnetic fields. Product highlight
OriginWhen charged particles move in electric and magnetic fields the following two laws apply:
where F is the force applied to the ion, m is the mass of the ion, a is the acceleration, q is the ionic charge, E is the electric field, and v x B is the vector cross product of the ion velocity and the magnetic field Using Newton's third law of motion yields: This differential equation is the classic equation of motion of charged particles in vacuum. Together with the particles initial conditions it determines the particle's motion in space and time. It immediately reveals that two particles with the same m/q behave the same. This is why the mass-to-charge ratio is an important physical quantity in those scientific fields where charged particles interact with magnetic (B) or electric (E) fields. ExceptionsThere are non-classical effects that derive from quantum mechanics such as the Stern–Gerlach effect that can diverge the path of ions of identical m/q. Symbols & UnitsThe official symbol for mass is m. The official symbol for electric charge is Q; however, q is also very common. Charge is a scalar property, meaning it can be either positive (+ symbol) or negative (- symbol). Sometimes, however, the sign of the charge is indicated indirectly. Coulomb is the SI unit of charge, however, other units are not uncommon. (note: The symbol "Q" is also used for other units of measure including flow.) The SI unit of the physical quantity m / q is kilogram/coulomb.
In the field of mass spectrometry the units and notation above are used in the physics of mass spectrometery while the unitless m/z notation is used as the independent variable that the mass spectrometer measures. These notations are closely related through the unified atomic mass unit and the elementary charge. See Mass spectrum. HistoryIn the 19th century the mass-to-charge ratio of some ions were measured by electrochemical methods. In 1897 the mass-to-charge ratio m / e of the electron was first measured by J.J. Thomson.[1] By doing this he showed that the electron, which was postulated before in order to explain electricity, was in fact a particle with a mass and a charge and that its mass-to-charge ratio was much smaller than that of the hydrogen ion H+. In 1898 Wilhelm Wien separated ions (canal rays) according to their mass-to-charge ratio with an ion optical device with superimposed electric and magnetic fields (Wien filter). In 1901 Walter Kaufman measured the relativistic mass increase of fast electrons. In 1913 J.J. Thomson measured the mass-to-charge ratio of ions with an instrument he called a parabola spectrograph.[2] Today, an instrument that measures the mass-to-charge ratio of charged particles is called mass spectrometer. See also
Further reading
References and notes
|
|||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Mass-to-charge_ratio". A list of authors is available in Wikipedia. |





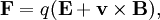 (Lorentz force law)
(Lorentz force law)
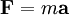 (Newton's second law of motion)
(Newton's second law of motion)