To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Molar absorptivityThe molar extinction coefficient, also known as molar absorptivity, is a measure of how strongly a chemical species absorbs light at a given wavelength. It is an intrinsic property of the species; the actual absorbance, A, of a sample is dependent on the pathlength l and the concentration c of the species via the Beer-Lambert law, A = εcl. The units of ε are usually in M-1cm-1 or L mol-1cm-1. Product highlightIn biochemistry, the extinction coefficient of a protein at 280 nm depends almost exclusively on the number of aromatic residues, particuarly tryptophan, and can be predicted from the sequence of amino acids.[1] If the extinction coefficient is known, it can be used to determine the concentration of a protein in solution. Another measure of the extinction coefficient is E 1% which gives the mass extinction coefficient. E1% is the absorbance of a 1% solution by mass and has the units g-1L cm-1. One can convert between ε and E1% using the following equation: ε=(E1%*molecular weight)/10.
The composition of a mixture of N components can be found by measuring the absorbance at N wavelengths (the values of ε for each compound at these wavelengths must also be known). The wavelengths chosen are usually the wavelengths of maximum absorption (absorbance maxima) for the individual components. None of the wavelengths must be an isosbestic point for a pair of species. For N components with concentrations ci and wavelengths λi, absorbances A(λi) are obtained:
This set of simultaneous equations can be solved to find concentrations of each absorbing species. References
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Molar_absorptivity". A list of authors is available in Wikipedia. |





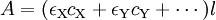 ,
,
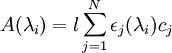 .
.


