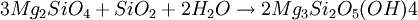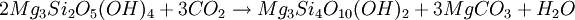To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Talc carbonateTalc carbonate is a geologic term for a suite of rock and mineral compositions found in metamorphic ultramafic rocks. The term refers to the two most common end-member minerals found within ultramafic rocks which have undergone talc-carbonation or carbonation reactions, talc and the carbonate mineral magnesite. Talc carbonate mineral assemblages are controlled by temperature and pressure of metamorphism and the partial pressure of carbon dioxide within metamorphic fluids, as well as by the composition of the rock. Product highlight
Fluid composition controlsTalc carbonation reactions occur in ultramafic rocks at lower greenschist through to granulite facies metamorphism when the rock in question is subjected to metamorphism and the metamorphic fluid has more than 10% molar proportion of carbon dioxide. When the metamorphic fluids in contact with the ultramafic rock have less than 10% CO2 the metamorphic reactions favor serpentnitisation reactions, resulting in chlorite-serpentine-amphibole type assemblages. Because it is practically impossible to know the carbon dioxide content of metamorphic fluids present during metamorphism, it is ofen difficult to assign a metamorphic grade to an ultramafic rock which has undergone talc carbonation because, for any given metamorphic assemblage, a rock could have been subjected to a range of temperature-pressure and fluid conditions. The metamorphic grade of an ultramafic rock is in most cases best understood by studying the rocks in contact with the ultramafic rocks. It is much easier to assign a metamorphic grade to a felsic rock, for example by the presence of biotite or garnet, because felsic metamorphic assemblages are essentially unaffected by carbon dioxide. Compositional controlsIn a general sense, talc carbonate metamorphic assemblages are diagnostic of the magnesium content of the ultramafic protolith.
Thus, the MgO content of a metamorphosed ultramafic rock can be estimated roughly by understanding the mineral assemblage of the rock. Magnesium content determines the proportion of talc and/or magnesite and aluminium-calcium-sodium content determines the proportion of amphibole and/or chlorite. Talc Carbonate MineralsSeveral minerals are diagnostic of talc carbonated ultramafic rocks;
At amphibolite facies, the diopside-in isograd is reached (dependent on carbon dioxide partial pressure) and metamorphic assemblages trend toward talc-pyroxene and eventually toward metamorphic olivine. Mineral ReactionsSerpentinisation of olivine Forsterite + aqueous silica → Serpentine Carbonation of serpentine to form talc-magnesite OccurrenceBecause carbon dioxide is such a common component of metamorphic fluids, talc-carbonated ultramafics are relatively common. However, the degree of talc-carbonation is usually somewhere between the two end-member compositions of pure serpentinite and pure talc-carbonate. It is common to see serpentinites which contain talc, amphibole and chloritic minerals in small proportions which infer the presence of carbon dioxide in the metamorphic fluid. Talc carbonate is present in many of the ultramafic bodies of the Archaean Yilgarn Craton, Western Australia. Notably, the Widgiemooltha Komatiite shows pure talc-carbonation on the eastern flank of the Widgeimooltha Dome, and almost pure serpentinite metamorphism on the western flank. Carbonation of other rocksCarbon dioxide has less severe impacts on mafic, felsic and rocks of other composition, such as carbonate rocks, chemical sediments, etcetera. The exception to this rule is the calc-silicate family of metamorphic rocks, which are also subjected to wide variations in mineral speciation due to the mobility of carbonate during metamorphism. Felsic and mafic rocks tend to be less affected by carbon dioxide due to their higher aluminium content. Ultramafic rocks lack aluminium, which allows carbonate to react with magnesium silicates to form talc. In rocks with extremely low aluminium contents, this reaction can progress to create magnesite. Advanced carbonation of felsic and mafic rocks, very rarely, creates fenite, a metasomatic alteration caused particularly by carbonatite intrusions. Fenite alteration is known, but very restricted in distribution, around high-temperature metamorphic talc-carbonates, generally in he form of a sort of aureole around ultramafics. Such examples include biotite-rich zones, amphibolite-calcite-scapolite alteration and other unusual skarn assemblages. See also
Categories: Metamorphic rocks | Igneous rocks |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Talc_carbonate". A list of authors is available in Wikipedia. |